
|
Journal of Applied Sciences and Environmental Management
World Bank assisted National Agricultural Research Project (NARP) - University of Port Harcourt
ISSN: 1119-8362
Vol. 9, Num. 1, 2005, pp. 45-49
|
Journal of Applied Sciences & Environmental Management,
Vol. 9, No. 1, 2005, pp. 45-49
Geochemical Implication of some Chemical Fossils as
Indicators of Petroleum Source Rocks
1*OSUJI,
L. C; 2ANTIA, B. S.
1Department
of Pure and Industrial Chemistry, University of Port-Harcourt, Port-HarcourtNigeria
2Department
of Chemistry, University of Uyo, Uyo Nigeria.
Code Number: ja05008
ABSTRACT:Chemical fossils (biomarkers)
are molecules in crude oils, source rocks and sediments whose carbon structures
or skeletons can be traced back to living organisms. Three of such diagnostic
fossils have been typically reviewed with respect to maturation changes,
precursor-product relationship, utility for source rock/crude oil, crude
oil/crude oil correlations and role in basin development. Fingerprints
(composition and distribution) of hopanes, oleananes and steranes have been
highlighted. Defunctionalization and breakage of double bonds which occur
during diagenesis as well as stereochemical changes which occur during
catagenesis have also been highlighted. Napthenic hydrocarbons, such as
steranes (C27 - C29) and triterpanes (C29 - C32),
are useful in geochemical correlations because of their unique compound
distributions and the fact that they are not seriously affected by migration,
thermal alteration and biodegradation. Although individual biomarkers are in
concentrations of only 10 to 200 ppm in crude oils, they can be accurately
measured in spite of their unusual complexity and variety which find use in
source rock correlation and maturation studies. Correlations using chemical
fossil technology can be applied in real cases by recognizing the source chromatogram
(GC) or fragmentogram of the hydrocarbon molecules in the oils to know whether
they have the same biomarkers or similar geohistory of origin and migration.
Therefore, genetically related oils are differentiated from unrelated oils on
the assumption that the same source material and environment of deposition
produce the same oil. Thus, a chemical fossil compound in a particular source
rock would be expected to appear in the oils it generated. @JASEM
The determination of the origin and fate of past life has
traditionally been the preserve of paleontologists, palynologists and
sedimentologists. They have relied on the morphology of macro, micro and trace
fossils to identify possible biotic precursors and infer the origin, and age of
the rocks in which the remnants of erstwhile living organisms are found (Tissot
and Welte,1984; Kruge,1986; Lo,1992; Margoon and Dow,1994). Regrettably, some
sedimentary rocks, for example, those containing mainly amorphous organic
matter (Types 1 and 11 kerogen) often contain few or no fossils, and besides,
these traditional methods cannot be applied to petroleum which being a fluid,
possesses neither macro- nor micro-fossils for visual or microscopic
examination. Hence, paleoreconstruction of cases such as these can only be
effectively undertaken at the molecular level by means of biomarkers, otherwise
called chemical-fossils (Pym, 1975; Petters, 1980; Philp and Gilbert, 1986;
Ekweozor and Telnaes, 1989; Moldowan et al., 1992; ).
Chemical fossils are molecules in crude oils, source rocks
and sediments whose carbon skeletons can be traced back to living organisms
(Hunt, 1996). These organic compounds which are unequivocally related to their
natural product precursors originated from chemical and geological
transformation of biomolecules of organisms that were deposited during
sedimentary processes. Within the stable carbon-carbon skeleton of such
compounds are embodied essential information on the habitat , nature and fate
of the ancestral flora and fauna which can facilitate the reconstruction of
environment of deposition of ancient sediments and petroleum. Such compounds
may also provide information on the thermal history of a basin or parts of a
basin (Ekweozor and Strauz, 1983; Kleme, 1989; Peters and Moldowan, 1993; Peters
and Cassa, 1994). The composition and distribution (fingerprint) of certain
diagnostic chemical fossil can therefore indicate the dominant source of
sedimentary organic matter (marine or non-marine), the physicochemical
conditions prevalent and the paleoenvironment (oxicity/anoxicity and salinity
status) as well as the maximum thermal stress experienced by the rocks or
petroleum in which the compounds are found (Staplin, 1979; Ekweozor and Strauz,
1982). Since petroleum is generated from the
organic matter in fine-grained rocks, it is possible to relate oil
accumulations in a stratigraphic column with specific source-rock intervals by
biomarker fingerprinting. Crude oils having a common source, but reservoired
at different horizons in a field or basin can likewise be correlated with one
another (Idowu and Ekweozor, 1989; Idowu et al., 1993; Hunt, 1996; Osuji and
Antia, 2002).
Typical chemical fossils include the
terpenoids (e.g., pristane, phytane, sterane, hopane); the porphyrins (e.g.,
chlorophyll), the odd-numbered n-paraffin carbon chains (e.g., C25
to C37) the iso- and anteiso- branched carbon chains (e.g.,
2-Methyldecane and 3-Methyldecane respectively) (Ekweozor, 1978; Ekweozor et
al., 1979a and b; Noble, 1991; Hunt, 1996).
Maturation Changes And
Precursor-Product Relationship
The thermal break-down of kerogen to form
oil during catagenesis results in significant changes in the biomarkers that
enable them to be used for source-rock evaluation. The predominance of the odd-carbon
normal-paraffin chains formed biologically is destroyed through the break-down
of larger equal amounts of even and odd chains generated thermally. Such
changes as defunctionalization and breakage of double bonds are common during
diagenesis (Fig. 1). In the hopane family (hopanoids) for example, the
hydroxyl groups are lost forming olefins; there is also breaking of double
bonds, but the ring system remains intact. Other prominent features of hopane
maturation include stereochemical changes at C-27, C-24 and C-29 and the
possibility of carbon-carbon cleavage. As seen in Fig. 2, these features occur
during catagenesis of the hopanoids where A (22R) is the least stable and C
(22R + S) the most stable. The abundance of R and S tells how close the geological
material is to the oil threshold (TIHG). Another typical example of maturation
changes is seen in the conformational transformation of the oleananes (Unomah,
1989). The oleananes have a biological conformation of a major 18β(H) and
minor 18α(H), and a geological
conformational inversion shows the precursor-product relationship in the
oleanoids, as seen in the schematic in Fig. 3. Apart from the hopanoids and
oleanoids, there are other typical examples of maturation changes in chemical
fossils, all of which show precursor-product relationship.These include
2,6,10,4-tetramethylpentadecane which has an original configuration of 6 (R),
10 (S) in the biosphere, but isomerizes into the 6 (S), 10 (S) and 6 (R), 10
(R) isomers in the geosphere giving a final mixture of the three configurations
in the ratios 2: 1: 1 for the 6 (S), 10 (R); 6 (S), 10 (S) and 6 (R)
configurations. For Steranes, transformations occur from 14α (H), 17α
(H): 20 (R) conformation to 14β (H), 17β (H): 20 (R+S) as final
conformation. These molecular or configurational transformations give an
insight into the degree of maturation, thermal stress that sediments have
passed through and other vital information necessary for paleoreconstruction.
(Simoneit, 1986; Ekweozor and Udo, 1987; Mango, 1990; Magoon and Dow, 1994;
Hunt, 1996).
Utility And Problems Of
Chemical Fossils
n- Alkane Distribution
The evaluation of the n-alkane distribution is a standard
method of classifying oils and rocks according to whether terrestrially or
marine-derived organic matter predominates in the source rock. Terrigenous
organic matter contributes to the high molecular weight usually with odd-even
carbon number predominance (OEP) in the range C25 to C35
and with maximization at C27, C29 or C31
alkanes. The Carbon Preference Index (CIP) and the Odd/Even Carbon Ratio (OER)
are both commonly used as a measure of maturity of organic matter and are
defined as follows:
C17 + C19
+ … + C29 + C31 C17 + C19
+ … C29 + C31
CPI = ½ +
- -- (1)
C16 + C18
+ … +C28 + C30 C18 + C20 +
… C30 + C32
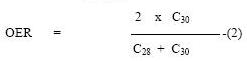
Both the CPI and OER in terrestrial organic matter decreases
from over 5.0 in recent sediments to 1.0 in mature sediments and oils as well
as in mature marine organic matter (Brassel et al., 1978)
Pristane/n-C17 vs. Phytane /n-C18 plot
and Pristane/Phytane Ratio
The nature of the organic matter and the redox potential in
the depositional environment during early diagenesis of the main precursor of
phytane and pristane, i.e., chlorophyll, or specifically its phytol chain, is
generally reflected by the ratio of Pristane (Pr) to Phytane (Ph). Marine
organic matter usually have Pr/Ph ratio < 1.5 while terrestrial O.M. have
ratios 3.0. Pr/Ph ratio therefore increases with increasing maturity. The
ratio Pr/n-C17 has been used to differentiate O.M. from swamp
environment (>1.0) from those formed under marine environment (<0.5), but
this ratio is affected by maturity, as well as biodegradation, hence the use of
a crossplot of Pr/n-C17 against Ph/n-C18.(Seifert, 1977;
Ekweozor, 1978; Ekweozor et al.,1981; Philp et al.,1983; Unomah, 1989;
Moldowan et al., 1992).
Steranes and Hopanes
The isomeric distribution of the steranes and diasteranes
have been used for assessing the maturity of source rocks. Naturally occurring
steroids and steranes from recent sediments have exclusively 5α(H) or 5β(H), 14α(H), 17α(H)
conformation while an increased proportion of the 20S isomer is generated with
maturity. Hence, the ratio, 20S / 20S + 20R - C29 sterane, has a
starting value of 0.0 in recent sediments, but progresses to an equilibrium mixture
achieved well into the oil window (TOGW). The onset of oil generation starts
at 0.4. Similarly, hopanes can be used for assessing the maturity of source
rocks. Thus, the ratio:
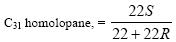
has a starting value of 0.0 and an equilibrium value of 0.6,
the complex isomerization at C-22 being achieved before the threshold of
intense hydrocarbon generation (TIHG).
Tricyclic and
Pentacyclic Terpanes
Abundant tricyclic terpanes have been detected in crude oil
and source rock extracts. Philp and Gilbert (1986) indicated that extended
tricyclic terpanes were abundant in marine sourced oils but generally absent in
terrigenous oils. Ekweozor et al (1979a and b) also reported that some
tricyclic terpanes were present in the Niger Delta oils and were derived from
sequential cleavage of the bonds of pentacyclic terpanes. Ekweozor and Strauz
(1982) identified some C19 - C30 tricyclic alkanes from a
hexaisoprenoid precursor. The distribution of pentacyclic triterpanes alone
have been very useful in source rock/crude oil correlations. Triterpane mass
chromatograms such as that of m/e 191 have been used to compare various crude
oils (Pym et al 1975; Hunt, 1996)
Chemical Fossils in Basin Development
The most relevant geochemical criteria are
those, which reflect the genetic relationship between organic matter at
different levels in the stratigraphic column as well as provide information
about migration of the mobile fluids. The data which they provide are useful
for source rock/crude oil, and crude oil/crude oil correlations which are
usually based on the recognition of compositional similarities. (Ekweozor et
al., 1976b; Tissot et al; 1974 Brassel et al; 1978). Correlation of crude oil
accumulations at different stratigraphic depths (horizons) in a well or in oil
fields located at different positions of a basin, is useful in determining the
importance of vertical and lateral migration in the area. This information,
when complemented with other geological criteria that indicate locations of
structures such as traps and faults, is useful in elucidating the most likely
migration patterns and hence would facilitate a more accurate siting of future
wells during basin development. (Brassel et al., 1978; Staplin, 1979; Ekweozor
et al, 1979a; Yukler, 1987; Hunt, 1996). Each family of oils represents one
element of a distinct petroleum system. Consequently, identifying the number of
oil families is equivalent to defining the number of petroleum systems. By
identifying the source rocks of each family, the drilling can focus on
prospects within the drainage areas of those sources (Cooper, 1990).
Application of Biomarker
Finerprints in Real Cases
Gas chromatograms or fragmentograms have
been widely used for correlating oils and source rocks since the pioneering
work of Seifert (1977) who differentiated oils produced from San Joaquin Basin
of California on the basis of sterane and terpane fingerprints. Recognizing
such source fingerprints of the hydrocarbon molecule enables us to know whether
they have the same biomarkers or similar geohistory of origin and migration.
Thus, genetically related oils can be differentiated from unrelated oils on the
assumption that the same source material and environment of deposition produce
the same oils in which case a chemical fossil compound in the source rock would
be expected to appear in the oils it generated. Obtaining a whole oil GC
fingerprint requires analyzing an entire oil for the C2 – C45
hydrocarbon range on a gas chromatograph with a fused silica capillary column.
Conclusion
The field of chemical fossil (biomarker) geochemistry is
fast growing with new discoveries invalidating or drastically modifying old
concepts. Chemical fossils have been found useful for both basin evaluation and
development. They constitute the most widely used correlation parameters for
comparing crude oils of different source, migration, and biodegradation, for
both crude oil – crude oil and crude oil – source rock correlations. They also
constitute the most useful method for comparing conventional oils with very
heavy oils, asphalts, and pyrobitumens. A more successful correlation, however,
is a multi-parameter approach using biomarkers (fingerprint and ratio),
isotopes, gas chromatograms, and the content of heavy metals, nitrogen,
sulphur, and the like. The study has opened new vistas in Earth Science namely
“Molecular Paleontology” and “Molecular Stratigraphy”, which are increasingly
being put to good use in geochemical exploration.
Nomenclature
C22= Hydrocarbon with 22 carbon atoms.
C-22 = Carbon No.22.
CPI = Carbon
Preference Index.
OER = Odd/Even Ratio.
OEP = Odd/Even
Predominance.
OM = Organic Matter.
Ph = Phytane.
Pr = Pristane.
TIHG = Threshold of
Intense Hydrocarbon Generation.
TOGW = Top of Oil Generation Window
Acknowledgements: This article was sourced from the authors’
unpublished seminar manuscripts in geochemistry vetted by Professor C. M.
Ekweozor of Getamme Geochem, Port Harcourt (formally of the Geochemistry
Research Group, University of Ibadan). The authors are grateful to him for
exposing them to the theory and practice of petroleum geochemistry. Many thanks
also to Chidi Enogwe of Mobil, Ehinola Ajayi of University of Ibadan and all
others who encouraged our spirit of workmanship.
REFERENCES
-
Brassel, S.G.,
Eglinton, G., Maxell, J. R. and Philip, R.D. (1978): “Natural background of
alkanes in the aquatic environment”. O. Hutzinger, I. H. Van Lelyveld and B.
C. J. Zoetman (eds.), Aquatic Pollutants. Pergamon Publishers Oxford 11
- 23.
-
Cooper,
B.S.(1990): Practical Petroleum Geochemistry. Robertson Scientific
Publications, London. Pp.40-48
-
Ekweozor, C.
M.(1978): “Organic geochemistry of some sediments in the Niger Delta: The
terpenoid aspect” Ph.D Thesis, University of Ibadan, Ibadan. Nigeria.
-
Ekweozor, C.
M., Okogun, J. I., Ekong, D. E. U. and Maxwell, J. R. (1979a):“Preliminary
organic geochemical studies of samples from the Niger Delta: I Analysis of
crude Oils for triterpanes”. Chem. Geol. 27, 11- 28.
-
Ekweozor, C. M.,
Okogun, J. I., Ekong, D. E. U. and Maxwell, J. R. (1979b):“Preliminary organic
geochemical studies of samples from the Niger Delta:11. Analysis of studies of
triterpane derivatives”. Chem Geol. 27, 29 - 37.
-
Ekweozor, C. M.
and Strauz, O. P.(1980): “18, 19-bisnor β(H)
–cheilanthane, a noveldegraded tricyclic sesterpenoid – type hydrocarbon from
Athabasca Oil Sands” Tet. Lett., 23,2711 - 14.
-
Ekweozor, C.
M., Okogun, J. I., Ekong, D. E. U. and Maxwell, J. R. (1981): “C24 – C27 degraded
triterpanes in Nigerian petroleum: Novel molecular markers of source/input
or organic maturation?” Journ.Geochem. Explor. 15, 653
– 662
-
Ekweozor, C. M.
and Strauz, O. P. (1983): “Tricyclic terpanes in the Athabasca oil sands:Their
geochemistry. In M. Bjoroy et al.(eds.), Advances in organic geochemistry
1981. Wiley , Chichester. Pp. 746-766
-
Ekweozor, C. M.
and Telnaes, N. (1989): Oleanane parameter: verification by quantitativestudy
of the biomarker occurrence in sediments of the Niger Delta. Org. Geochem.,16
(1-3), 401-413
-
Ekweozor, C. M.
and Udo, O.T. (1987): “The Oleananes, origin, maturation and limits of
occurrence in Southern Nigeria sedimentary basin”, In Mattarelli, I. and
Nivelli, L. (eds.), Advances in Organic Geochemistry. Pergamon Press, Oxford
-
Hunt, J. M.
(1996): Petroleum Geochemistry and Geology. W.H. Freeman and Co., San
Francisco pp. 301 – 322.
-
Idowu J. O. and
Ekweozor,C. M (1989): “Petroleum geochemistry of some UpperCretaceous shales
from the Upper Benue Trough and Southwestern Chad Basin, Nigeria” Journ. Min.
Geol. 25, 131 – 150
-
Idowu, J. O.,
Ajiboye, S. A., Ilesanmi, M. A. and Tanimola, A. (1993): “Origin
andsignificance of organic matter of Oshosun formation, Southeastern Dahomey
Basin, Nigeria”, Journ. Min. Geol. 29(1), 9 – 17
-
Klemme, H. D.
(1994): “Petroleum systems of the world that involve Upper Jurasic source
rocks” In L. B. Magoon and W. G. Dow (eds.), The petroleum system: From source
to trap. AAPG Memoir 60. Published by American Association of Petroleum
Geologists, Tulsa. Pp. 51 – 72
-
Kruge, M. A.
(1986): “Biomarker geochemistry of the Miocene Monterey formation,West San
Joaquin Basin California: implications for petroleum generation” Organic
Geochem. 10, 517 – 530
-
Lo, H. B.
(1992): “Identification of indigenous vitrinites for improved thermal maturity
evaluation” Organic Geochem. 718 (3), 339 –364.
-
Mango, F. C.
(1994): “The origin of light hydrocarbons in petroleum: ring preference in the
closure of carboxylic rings”. Geochim. Cosmochim. Acta. 58(2), 895 – 901
-
Magoon, L. B.
and Dow, W. G.(1994): Eds., The petroleum system – From source totrap. AAPG
Memoir 60, published by American Association of Petroleum Geologists, Tulsa.
Pp. 3 – 24
-
Moldowan, J.
M., Albrecht, P. and Philp, R. P. (1992): Eds., Biological Markers in Sediments
and Petroleum. Prentice – Hall, Englewood Cliffs, NJ. Pp. 12 – 26
-
Noble, R. A.
(1991): “Geochemical techniques in relation to organic matter”. In R. K.Merrill
(ed.), Source and migration techniques. American Association of Petroleum Geologists,
Tulsa. Pp.97 - 102
-
Osuji, L. C.
and Antia, B. S. (2002): “An infra-red spectroscopic evaluation of the
petroleum potentials of some oil shales from Lokpanta in the Lower
Benue Trough of Nigeria” J. Appl. Sci. Environ. Mgt. 6(1), 34 – 38
-
Peters, K. E.
and Cassa, M. R. (1994): “Aplied source rock geochemistry” In L. B. Magoon and
W. G. Dow (eds.), The petroleum systems – From source to trap. AAPG Memoir 60,
published by American Association of Petroleum Geologists, Tulsa. Pp. 93 – 120
-
Peters, K. E.
and Moldowan, J. M. (1993): The biomarker guide: Interpreting molecular
fossils in petroleum and ancient sediments. Prentice – Hall, Englewood
Cliffs, NJ
-
Petters, S. W
(1980): “Biostratigraphy of Upper Cretaceous Formaminifera of the Benue Trough Nigeria” Journ.
Foram.Research, 10, 191 - 204.
-
Philp, R. P.
and Gilbert, T. D. (1986): “Biomarker distribution in Australian
oilspredominantly derived from terrigenous source material”. Org. Geochem. 10,
73 – 84
-
Philp, R. P.,
Simoneit, B. R. T. and Gilber, T. C. (1983): Diterpenoids in crude oils
andcoals of southern Australia. In M. Bjoroy et al.(eds.), Advances in organic
geochemistry 1991. Wiley, Chicheser. Pp.698 - 704
-
Pym, J. G.,
Ray, J. E., Smith, G. W. and Whitehead, E. V. (1975): “Petroleum triterpane
fingerprinting of crude oils”.Anal. Chem. 47, 1617 - 22.
-
Seifert, W. G.
(1977): “Source rock/oil correlations by C27 - C30 biological
markerhydrocarbons”. Proc., Seventh Intern. Meeting on Organic Geochem,
Madrid. Pp. 7 - 12
-
Simoneit, B. R.
T. (1986): “Cyclic terpenoids of the geosphere”. In R. B. Johns (ed.),
Biological Markers in the Sedimentary Record: Methods in Geochemistry and
Geophysics 24. Elsevier Science, Amsterdam. Pp. 43 – 49
-
Staplin, F. L.
(1979): “Sedimentary organic matter, organic metamorphism and oil and gas
occurrence”. Canadian Petrol. Geol. Bull. 19 (1) 47 - 66.
-
Tissot, B. P.,
Durand, B., Espitalie, J. and Combaz, A. (1974): “Influence of nature
anddiagenesis of organic matter in formation of petroleum Amer. Asso. Petrol.
Geol. Bull. 58, 499 - 506.
-
Tissot, B. P.
and Welte, D. H (1984): Petroleum formation and occurrence, a new approach
to oil and gas exploration. N.Y. Springer - Verlag, Berlin. pp. 500 -
38.
-
Unomah, G. I.
(1989): “Petroleum evaluation of Upper Cretaceous shales in Lower Benue Trough,
Nigeria” Ph. D. Thesis, Univ. of Ibadan, Ibadan Nigeria
-
Yukler, M. A.
(1987): Role of organic geochemistry in quantitative basin analysis. In
L.Mattavelli and L. Novelli (eds.), Advances in organic geochemistry, Pergamon
Press, Oxford. Pp. 126 - 142
Copyright 2005 - Journal of Applied Sciences & Environmental Management
|
