
|
Memórias do Instituto Oswaldo Cruz
Fundação Oswaldo Cruz, Fiocruz
ISSN: 1678-8060 EISSN: 1678-8060
Vol. 99, Num. 5, 2004, pp. 443-470
|
Mem Inst Oswaldo Cruz, Rio
de Janeiro, Vol. 99, No. 5, August, 2004, pp. 443-470
Blackflies (Diptera: Simuliidae)
of Southern Guyana with Keys for the Identification of Adults and Pupae
- A Review
AJ Shelley/+,
LM Hernández, JB Davies*
The Natural History
Museum, Department of Entomology, Cromwell Road, London SW7 5BD, UK *Liverpool
School of Tropical Medicine, Liverpool, UK
+Corresponding
author. Fax: +44-207-942.5661. E-mail: ajs@nhm.ac.uk
Received 12 January
2004
Accepted 19 May
2004
Code Number: oc04101
A revision is
made of the previously poorly studied blackfly fauna from the south-western
border of Guyana with Brazil. Notes on the biosystematics of the species
found are provided, together with keys and illustrations based on their
morphology. Of the 14 species recorded, eight are anthropophilic and two
of these (Simulium oyapockense s.l. and S. guianense s.l.) are
proven vectors of human onchocerciasis in the nearby Amazonia focus of
the disease in neighbouring Brazil.
Key words: blackflies
- Simuliidae - Neotropical region - taxonomy - Guyana
There have been
few studies on the Simuliidae in Guyana and all those that have been made
involve the border area with Brazil in the Rupununi District. This has largely
been due to the lack of simuliids in the swampy coastal regions, the difficulty
of access to the interior of the country and the lack of any pathogenic parasites
transmitted by these insects. It was in Guyana (then British Guiana) at the
end of the last century that relatively non pathogenic, blood inhabiting
microfilariae were sent by Dr Ozzard to Manson, who described them as the
new species Mansonella ozzardi. It was soon discovered that this filarial
species was common in the Caribbean Islands where the vectors were species
of Culicoides midges. Eighty years later the discovery of simuliids
as vectors of M. ozzardi in Brazil and Colombia (reviewed in Shelley & Coscarón
2001) prompted the work of Nathan et al. (1982), who discovered that Simulium
oyapockense s.l. is a vector of M. ozzardi in the Rupununi
District of Guyana.
At the beginning
of the twentieth century studies on anthropophilic Diptera were numerous
because of their biting nuisance and, in the case of mosquitoes, their proven
role as vectors of pathogenic species of protozoal parasites. The first attempt
at study of the Simuliidae in Guyana was made by the government Surgeon General
at Georgetown, KS Wise. He had been sent man-biting simuliids by Melville
from the upper branches of the Essequibo river (R) (the Rupununi and Siparuni
R) and also examined specimens of two species from the same area in the Museum
of the Royal Agricultural and Commercial Society of British Guiana in Georgetown.
Wise (1911) provided a redescription for one of the species known as the "pium" in
Brazil [by the Macuxi indians] and the "cabouri" fly by the Arawak
indians in Guyana, and which had already been described in Brazil [by Lutz]
as S. amazonicum. This species is not S. amazonicum but S.
oyapockense s. l., a species later described in the 1940s from
French Guiana (Shelley et al. 1997) and also known in Brazil as the "pium".
Wise described the other species as new, S. guianense, which the local
indians referred to as the itanimi fly to distinguish it from the smaller
pium or cabouri fly. In 1915 Knab described the anthropophilic female, sent
to him by Dr Wise from the same area, as the new species S. limbatum.
At a time when
it was considered that any development of the interior of the country would
need to take account of blackfly control, Drs OW Richards and J Smart visited
Guyana in 1937. They recorded seven simuliid species from this country and
published taxonomic notes and a key to these, as well as other species collected
from the Lesser Antilles (Smart 1940). The seven species from Guyana were: S.
amazonicum (= S. oyapockense s.l.), S. guianense (= S.
perplexum), S. haematopotum (= S. oyapockense s.l.), S.
limbatum, S. lutzianum (= S. kabanayense), S. rubrithorax (= S.
maroniense s.l.), S. sanguineum (= S. oyapockense s.l.).
It is now known that these names represent only five distinct species as
indicated in brackets. In most cases the misidentifications are completely
understandable because at this time many species were poorly defined morphologically.
However, an obvious lapse occurred with pupae identified as S. rubrithorax (with
eight gill filaments), which are S. maroniense s.l. (with 18-22 gill
filaments). Similarly, the naming of three pupal exuviae (in BMNH collection)
as S. lutzianum s.l. (with eight gill filaments) was incorrect
as the specimens have 10-13 filaments and are of a species later described
as S. kabanayense (Ramírez Pérez & Vulcano 1973).
These three specimens were not referred to in the taxonomic discussion of S.
lutzianum s.l. in Shelley et al. (1997) because they were known
not to be of this species but were not identified at the time. In 1989b Shelley
et al. described the new species S. perplexum based on specimens identified
by Smart (1940) as S. guianense Wise.
The collections
by the third author, which have been the most comprehensive for the country,
were made in the 1970s when the development of the Rupununi savanna for tourism
was being contemplated. One of the constraints to this development was the
presence of large numbers of anthropophilic simuliids. This work was necessary
for the development of studies on the biology of and a pilot control project
for S. limbatum (as S. in-crustatum) and S. oyapockense
s.l. (as S. amazonicum/S. sanguineum, S. amazonicum or S. sanguineum complexes)
(Humphrys et al. 1977, Rambajan 1981a, b). At this time the taxonomy of the
Simuliidae of this region of Latin America was relatively unknown, and it
was not until the finding of onchocerciasis in the Brazil-Venezuela Amazonia
focus some 400km to the west of the Rupununi District that comprehensive
taxonomic studies began.
This paper, based
on holdings at the Natural History Museum, London (BMNH) mainly including
the specimens of Wise, Knab, Smart, and the third author, provides the first
modern review of the Simuliidae of Guyana, albeit limited to the south-western
frontier area of the country with Brazil. Illustrated keys to the adults
and pupae of the 14 species of Guyana are presented, together with biosystematic
notes for each species and a list of material examined.
MATERIALS
AND METHODS
Collections were
made in 1970 and 1975 in three savanna areas of the Rupununi District on
the western border of Guyana with the state of Roraima Brazil (Map). Two
are situated in highland areas (400-450 m) around Orinduik on the R Ireng
and Kato on the R Chiung, and the other farther south in a more lowland area
(145 m) around Lethem on the R Takutu. Rivers, streams (creeks) and drainage
channels in these areas were prospected for immature stages of simuliids
from which mature pupae were selected and reared to adults, and man-biting
females were also collected. Standard techniques for capture, dissection
and conservation of material have already been described in Shelley et al.
(1997). All material has been deposited in the Department of Entomology of
the BMNH. Time constraints have meant that only reared and man-biting specimens
have been used in this paper and they are listed under Material Examined.
These data, together with all the original collecting data, are stored at
the NHM on a CD. Further material has been examined from Clemson University
Arthropod Collection, Clemson University, South Carolina, US (CUAC), Instituto
Nacional de Pesquisas da Amazônia, Manaus, Amazonas, Brazil (Inpa)
and the Institut Pasteur, Paris, France (IP). Figures of simuliid morphology
have been obtained from a Synoptics image capture system as detailed in Shelley
et al. (2000). Identification of material has been largely based on papers
describing the Venezuelan fauna (Ramírez Pérez 1983) and the
Brazilian Amazonia onchocerciasis focus fauna (Shelley et al. 1997), as well
as the papers cited in the text. Recent publications from neighbouring French
Guiana (Hamada & Fouque 2001) and Venezuela and Brazil (Hamada & Grillet
2001) also provide an insight into simuliid species that may be present in
Guyana if more comprehensive surveys are to be carried out in the future.
CHECKLIST
OF THE SIMULIIDAE OF GUYANA
Crosskey and Howard's
world inventory of the Simuliidae (1997) records 13 species of simulids from
Guyana: S. cauchense, exiguum s.l., guianense s.l., haematopotum, incrustatum,
limbatum, oyapockense s.l., perflavum, perplexum, pulverulentum, quadrifidum,
rorotaense, and S. rubrithorax. Since this publication the taxonomy
of the species of the Amazon and surroundings has advanced and so the following
comments can be made. The records of S. rubrithorax Lutz and S. haematopotum Malloch
date back to Smart (1940). Their correct identifications (see Shelley et
al. 1997, 2002) are S. maroniense s.l. Floch & Abonnenc and S.
oyapockense s.l. Floch & Abonnenc, respectively. Simulium pul-verulentum Knab
was based on a record in Vulcano (1981), but Shelley et al. (2002) in their
revision of this species discounted its presence in Guyana based on lack
of information on specimens and the known distribution of this species. The
record for S. incrustatum Lutz refers to the closely related S.
limbatum Knab (see Shelley et al. 1997 for morphological diagnoses).
The origin of the record of S. exiguum Roubaud cannot be traced,
but the present work confirms its presence in the area studied (see Material
Examined). Also S. rorotaense should read as S. maroniense s.l. Consequently,
prior to the present paper only nine simuliid species had been reliably recorded
from Guyana. The 14 species now known from the country are listed in alphabetical
order by subgenera and by species within each subgenus. Five species, S.
clarki, S. kabanayense, S. lutzianum s.l., S. spinibranchium, and S.
subpallidum represent new country records. Species marked with an asterisk
bite man.
Simulium (Chirostilbia) spinibranchium Lutz,
1910 *
Simulium (Chirostilbia) subpallidum Lutz,
1910
Simulium (Inaequalium)
clarki Fairchild, 1940 *
Simulium (Notolepria)
exiguum Roubaud, 1906 (species complex) *
Simulium (Psaroniocompsa) cauchense Floch & Abonnenc,
1946
Simulium (Psaroniocompsa)
limbatum Knab, 1915 *
Simulium (Psaroniocompsa)
oyapockense Floch & Abonnenc, 1946 (species complex)
*
Simulium (Psaroniocompsa)
quadrifidum Lutz, 1917
Simulium (Psilopelmia)
kabanayense Ramírez Pérez, 1973
Simulium (Psilopelmia) lutzianum Pinto,
1932 (species complex)
Simulium (Psilopelmia)
maroniense Floch & Abonnenc, 1946 (species complex)
Simulium (Psilopelmia) perflavum Roubaud,
1906
Simulium (Trichodagmia)
guianense Wise, 1911 (species complex) *
Simulium (Trichodagmia) perplexum Shelley,
Maia-Herzog, Luna Dias & Couch, 1989 *
[It is probable
that the anthropophilic species S. roraimense Nunes de Mello, 1974,
also exists in Guyana - See "Notes on the Biosystematics of Species" under S.
oyapockense s.l.]
KEYS
TO THE SIMULIIDAE OF GUYANA
Morphological characters
that are usually observed in un-dissected material were used wherever possible.
Scutal patterns described are those seen in specimens placed with their bodies
parallel to the microscope stage and with the fibre optic light source immediately
in front of the head of the specimen. For the effects of light source direction
on scutal patterns see Shelley et al. (1997). Where a species complex occurs
the species name is followed by s.l. [sensu lato]. S. kabanayense was
only found in Guyana as pupae and consequently this species has not been
included in the adult keys. A description of these stages in this orange
species is found in Ramírez Pérez and Vulcano (1973).
FEMALES
1. Thorax orange
.................2
- Thorax grey or black
.........5
2 . Thoracic pattern
consisting of four, wide, faintly pruinose longitudinal lines; lateral scutal
borders greyish (Fig. 2) .......................................................................................................subpallidum (orange
form)
- Thorax without a
pattern; lateral scutal borders silver pruinose .......3
3. Humeri orange
or yellowish, never pruinose (Fig.10). Paraprocts
sub-quadrangular (Fig. 21).................. lutzianum
s.l.
- Humeri orange or
yellowish with silver pruinosity in some light incidences (Figs.
11,12). Paraprocts sub-triangular (Figs
22, 23).....................................................
4
4. Abdominal tergites
shiny black, first two segments silver pruinose ...................................maroniense
s.l.
Abdominal tergites
brown and orange, mottled with no pruinose areas................................ perflavum
5. Scutum without
pattern...................................................6
- Scutum with pattern
........................................................8
6. Scutum with
groups of silvery gold setae appearing green in some lights and arranged
in lines diverging from median line (Fig.
5). Nudiocular area absent (Fig. 27) .............................................................exiguum
s.l.
- Scutum with
groups of cream or brass-coloured, fine or scale-like setae in
at least anterior two thirds and not arranged in diverging lines
(Figs 13, 14). Nudiocular area
present (Figs 30, 31).......................7
7. Scutal setae
arranged irregularly in groups in anterior two thirds and densely and individually
on posterior third (Fig. 13). Basal section
of radius of wing with single row of setae (Fig.
32). Claws of hind leg without basal teeth (Fig.
34). Paraproct broadly quadrangular with small, internal, membranous
extension including anterior tail-like projection (Fig.
24); genital fork with distal ends of lateral arms well developed with
small anterior processes (Fig. 40) ...............guianense
s.l.
- Scutal setae
arranged regularly in small groups, except singly and sparsely
on posterior border (Fig. 14).
Basal section of radius of wing bare (Fig.
33). Claws of hind leg with basal teeth (Fig.
35). Paraproct broadly rectangular with internal extension
well developed, not membranous and lacking anterior tail-like
projection (Fig. 25); genital
fork with distal ends of lateral arms less developed with larger
anterior processes (Fig. 41).......................................................... perplexum
8. Scutal pattern
largely black with 1+1 sub-median, posteriorly diverging, silver pruinose
lines of varying length (Figs 4, 6, 7)......................................................................................
9
- Scutal pattern
almost equally black and silver pruinose, or mainly silver pruinose
(Figs 1, 3, 8,
9) ........................11
9. Nudiocular area
absent (Fig. 28). Cibarium armed with blunt
tubercles (Fig. 45)..........................................cauchense
- Nudiocular area
absent or well developed (Figs 26,
29). Cibarium armed with sharply pointed teeth (Figs
44, 46)......10
10. Cibarial teeth
present along anterior margin, including median protuberance, cornuae well
developed (Fig. 44). Paraproct well developed,
extending beyond ventral edge of cercus by length of cercus (Fig.
17)............................clarki
- Cibarial
teeth present along anterior margin, except for area of median
concavity, cornuae poorly developed (Fig.
46). Paraproct small, only slightly protruding beyond ventral
edge of cercus (Fig. 18)............................................... limbatum
11. Scutal pattern
composed of black and silver, longitudinal pruinose areas in almost equal
proportions (Figs 8, 9). Cibarium armed with
teeth or blunt tubercles (Figs 47, 48). Paraprocts
poorly developed with little extension beyond ventral surface of cercus (Figs
19, 20)...........................................................................12
- Scutal pattern
composed of longitudinal, largely pruinose bands and narrow black
bands (Figs 1, 3). Cibarium unarmed
(Figs 42, 43). Paraprocts well
developed with triangular extension beyond ventral margin of
cerci (Figs 15, 16).............13
12. Cibarium with
rows of sharp teeth along anterior margin (Fig.
47). Genital fork lightly sclerotised with poorly developed terminations
to lateral arms (Fig. 38)........................................oyapockense
s.l.
- Cibarium with blunt
tubercles on anterior margin (Fig.
48). Genital fork well sclerotised with well developed terminations
to lateral arms (Fig. 39)........................................................................... quadrifidum
13. Scutum with
1+1 longitudinal, sub-median bands wide and diverging anteriorly (Fig.
1); scutellum greyish black. Hind legs with distal part of femora yellow
and of tibiae dark brown (Fig. 36)...............................................spinibranchium
- Scutum with 1+1
longitudinal, sub-median bands converging anteriorly (Fig.
3); scutellum yellowish orange. Hind leg yellowish with distal
articulations of femora and tibiae dark brown (Fig.
37) ...............................subpallidum (dark
form)
MALES
1.Thorax orange
.......................................................2
- Thorax grey or black
..............................................5
2. Scutum with
1+1 large, silver pruinose patches on anterior third of scutum beginning
at anterior margin [best seen when specimen tilted] (Fig.
50). Gonostyle sub-triangular with no terminal spine, shorter than gonocoxite
(Fig. 66); ventral plate wider than long with
poorly developed keel and basal arms (Fig. 79)...................................... subpallidum (orange
form)
- Scutum without pattern
(Figs 60-62). Gonostyle elongate
with prominent terminal spine, as long as gonocoxite (Figs
73-75); ventral plate as wide as long or longer than wide,
with or without well developed keel and with well developed inwardly
curved basal arms (Figs 86-88)
...............................................................................3
3. Keel of ventral
plate slightly developed (Fig. 86). Legs distinctly
dark brown except for most of basitarsi of mid and hind legs, which are yellow
(Fig. 93)........................................................ lutzianum
s.l.
- Keel of ventral
plate well developed (Figs 87, 88).
Legs distinctly yellowish or pale brown, including basitarsi
of mid and hind legs (Figs
94, 95).............................................................................
4
4. Keel of ventral
plate with upright hairs (Fig. 87). Sc of
wing with setae (see Fig. 32 for location
of vein Sc on wing) .....................................................................................maroniense
s.l.
- Keel of ventral
plate with adpressed hairs (Fig. 88).
Sc of wing without setae ..........................................perflavum
5. Scutum without
pattern ................................................6
- Scutum with pattern
......................................................9
6. Scutum with
setae short and arranged in small packets or groups (Figs
53, 64) .....................................7
- Scutum with setae
long and not arranged in small packets or groups (Figs
54, 58)..................................
8
7. Gonostyle conical
and shorter than gonocoxite (Fig. 68); ventral
plate wider than long with triangular keel and reduced basal arms (Fig.
81) ...................................................exiguum s.l.
- Gonostyle sub-rectangular
and as long as gonocoxite (Fig. 77);
ventral plate wider than long with concavity in place of keel
and well developed basal arms (Fig.
90) ..............................perplexum
8. Paramere with
poorly developed spines (Fig. 91) .....................................cauchense
- Paramere with well
developed spines (Fig. 92) ...........................................quadrifidum
9. Scutal pattern
in anterior third of scutum consisting of 1+1 divergent, well defined, sub-median
cunae or vittae (Figs 55-57, 59). Ventral
plate sub-triangular or arcuate (Figs 82-85)
..............................10
- Scutal pattern consisting
of extensive areas of white pruinosity in anterior third of scutum
or black and white pruinose pattern involving whole scutum (Figs
49, 51, 52, 63). Ventral plate
sub-triangular (Fig. 80), arcuate
(Figs 78, 79) or H shaped (Fig.
89) ...............................................................................................13
10. Scutum with
sub-median cunae (Figs 56, 57). Gonostyle
sub-quandrangular or conical with central or terminal spine and considerably
shorter than gonocoxite (Figs 70, 71) ....................................11
- Scutum with sub-median
thin vittae (Figs 55, 59). Gonostyle
conical with terminal spine and about as long as gonocoxite (Figs
69, 72) .....................................................12
11. Sub-median
cunae each with smaller black cuna within (Fig.
56). Gonostyle sub-quadrangular with central spine (Fig.
70) ...................................................limbatum
- Sub-median cunae
with no internal black cunae (Fig.
57). Gonostyle conical with terminal spine (Fig.
71) ........................................oyapockense
s.l.
12. Paramere with
poorly developed spines (Fig. 91) ..................................................cauchense
- Paramere with well
developed spines (Fig. 92) ..........................................................quadrifidum
13. Scutal pattern
consisting of thick black median vitta extending for three fourths scutal
length and 1+1 lateral, wide, black vittae in middle portion of scutum (Fig.
63). Gonostyle conical with terminal spine and twice as long as gonocoxite
(Fig. 76); ventral plate H-shaped with central
promininence (Fig. 89) .........................................guianense
s.l.
- Scutal pattern consisting
of extensive areas of white pruinosity in anterior third of scutum
(Figs 49, 51, 52). Gonostyle conical
or sub-quadrangular, with or without terminal spine and as long
as or shorter than gonocoxite (Figs
65-67); ventral plate triangular or arcuate (Figs
78-80) ........................................................14
14. Scutal pattern
of 1+1, sub-median, broad, white pruinose bands on anterior scutal margin
(Fig. 52). Gonostyle conical with terminal
spine, shorter than gonocoxite (Fig. 67);
ventral plate triangular (Fig. 80)....................................... clarki
- Scutal pattern consisting
of white pruinose band along anterior scutal margin (Figs
49, 51). Gonostyle sub-quadrangular or conical with no spine
and shorter than gonocoxite (Figs
65, 66); ventral plate arcuate (Figs
78, 79) ...............15
15. Gonostyle sub-quadrangular,
less than half as long as gonocoxite (Fig. 65)
.................................spinibranchium
- Gonostyle conical,
almost as long as gonocoxite (Fig.
66) ......................................................subpallidum (dark
form)
PUPAE
1. Gill with 4
to 8 filaments .............................................2
- Gill with 10 or
more filaments ......................................11
2. Gill with 4
filaments ....................................................3
- Gill with 6 to 8
filaments ..............................................4
3.Primary branching
of gill in vertical plane; gill branch bifurcations usually not at same level
at mid point of gill (Fig. 100). Abdominal
tergite V without spines or spines combs .............................................................cauchense
- Primary branching
of gill in horizontal plane; gill branches usually bifurcate
at same level in basal 1/5 of gill (Fig.
103). Abdominal tergite V with weak sub-median row of spines
(usually present) along anterior border terminating in weak patch
of spines combs at margins .........................................................................................quadrifidum
4. Gill with 6
filaments ...................................................5
- Gill with 8 filaments
.....................................................7
5. Gill approximately
half length of pupa (gill length - 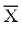 =
1.0 mm, range 0.8-1.5 mm, n = 10; pupa length - =
1.0 mm, range 0.8-1.5 mm, n = 10; pupa length -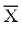 = 1.9 mm, range 1.5-2.1 mm,
n = 10*), with basal branching and
gill trunk dividing into three primary branches (Fig.
102) .........................................................................................oyapockense
s.l. = 1.9 mm, range 1.5-2.1 mm,
n = 10*), with basal branching and
gill trunk dividing into three primary branches (Fig.
102) .........................................................................................oyapockense
s.l.
- Gill almost
as long as or longer than pupa, gill branching varying from basal
to most distal at mid point of gill and gill trunk dividing into
two or three primary branches (Figs
98, 101) .............................................6
6. Gill slightly
longer than pupa (gill length - 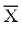 =
3.3 mm, range 2.4-4.1 mm, n =10; pupa length - =
3.3 mm, range 2.4-4.1 mm, n =10; pupa length -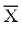 = 2.9 mm, range 2.2-3.5 mm,
n = 10); gill branching basal and
gill trunk dividing into three primary branches (Fig.
98) ...........................clarki = 2.9 mm, range 2.2-3.5 mm,
n = 10); gill branching basal and
gill trunk dividing into three primary branches (Fig.
98) ...........................clarki
- Gill almost as long
as pupa (gill length - 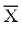 =
2.4 mm, range 2.1-2.8 mm, n = 10; pupa length - =
2.4 mm, range 2.1-2.8 mm, n = 10; pupa length -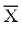 = 2.6 mm, range
1.8-3.6 mm, n = 10); gill branching varying from
basal to most distal at mid point of gill and gill trunk dividing
into two primary branches (Fig. 101)........................................................................limbatum = 2.6 mm, range
1.8-3.6 mm, n = 10); gill branching varying from
basal to most distal at mid point of gill and gill trunk dividing
into two primary branches (Fig. 101)........................................................................limbatum
7. Gill filaments
broadest at base and becoming progressively finer towards tip, thereby resembling
stag's antler (Fig. 96) .........................................................................spinibranchium
- Gill filaments narrow
along entire length.......................................................8
8. Gill filaments
arranged in bunch with all secondary branches arising very near gill base
(Fig. 108) .................perflavum
- Gill filaments not
arranged in a bunch with only some secondary branches arising
very near gill base (Figs 97, 99, 104)........................................
9
9. Gill trunk bifurcating
near base (Fig. 104).................................................lutzianum
s.l.
- Gill trunk dividing
into three primary branches near base (Figs
97, 99)................................ 10
10. Cocoon with
closely meshed fibres (Fig. 113). Trichomes
of frontoclypeus bifid to 5-branched and tubercles sparsely distributed (Fig.
115) ....................................................subpallidum
- Cocoon with open
weave (Fig. 114). Trichomes of
frontoclypeus simple or bifid (rarely with 3 branches) and tubercles
densely distributed (Fig. 116)
.......................................exiguum s.l.
11. Gill with 10-14
filaments (Figs 105, 106, 109,
111)............................................... 12
- Gill with 17-23
filaments (Fig. 107)............................................................................ maroniense
s.l.
12. Gill with
10-14 long, fine filaments rounded apically; gill trunk branching basally
into 10 primary branches, some of which may have secondary bifurcations (Figs
105, 106).................................................... kabanayense
- Gill with 12 short,
pointed filaments; gill trunk branching basally to form four
primary branches (Figs 109, 111)..........
13
13. Gill filaments with annulations
on distal part of gill less accentuated with spicules (Fig.
110).................. guianense s.l.
- Gill filaments with
annulations on distal parts of gill accentuated by forwardly
directed processes (Fig. 112)............................ perplexum
*data from Shelley
et al. (1997)
NOTES
ON THE BIOSYSTEMATICS OF SPECIES
Simulium (Chirostilbia)
spinibranchium Lutz
This species is
dealt with in Coscarón (1991), Py-Daniel & Shelley (1980) and
Shelley et al. (2000). It is an uncommon highland species previously only
recorded from Brazil and Southern Venezuela. In Guyana (see Material
Examined) it was collected in the mountainous area of the Orinduik region
near the R Ireng. Pupae were collected in small waterfalls, and on rocks
covered with red algae and attached to dead leaves. It has been collected
at similar sites on the Brazilian side of the R Ireng (R Maú in Brazil)
where females voraciously bite man (AJ Shelley & APA Luna Dias, unpublished
data).
Simulium (Chirostilbia) subpallidum Lutz
A full description
is given by Coscarón (1991) and Shelley et al. (2000); the latter
authors summarise the relevant literature. Over its range this species varies
in scutal coloration from faded orange to light grey. Black specimens were
also recorded in the present work. It is common and widespread throughout
neighbouring Brazil and also occurs in Venezuela, Argentina, and Paraguay.
Pupae were collected on vegetation in rivers 3-50 m wide, with rocky beds,
in the Rupununi District.
Simulium (Inaequalium)
clarki Fairchild
Since the original
description by Fairchild (1940) based on specimens from Panama the species
has had no full taxonomic treatment. It has also been recorded from Brazil
and Venezuela and now in the northern Rupununi District of Guyana, where
it was anthropophilic and found breeding in 50 m wide rivers. In the neighbouring
part of Brazil it was also collected biting man and found breeding in smaller
streams (AJ Shelley & APA Luna Dias, unpublished data).
Simulium (Notolepria)
exiguum Roubaud
(species complex)
Shelley et al.
(1997, 2000) reviewed the taxonomy of this widespread, commonly occurring
Neotropical species. Many populations of this species complex are anthropophilic
and some are vectors of human onchocerciasis, but in some areas almost total
zoophily occurs. S. exiguum s.l. has been collected breeding on vegetation
in the R Takutu (R Tacutu in Brazil) on the border between Brazil and Guyana.
Simulium (Psaroniocompsa)
cauchense
Floch & Abonnenc
This zoophilic
species has been reviewed in Shelley et al. (1997) and occurs in Brazil,
French Guiana and Venezuela. In Guyana S. cauchense was collected
in 15-50 m wide rivers, attached to leaves and submerged vegetation.
Simulium (Psaroniocompsa)
limbatum Knab
S. limbatum is
a voracious, anthropophilic species occurring in the Guianas, Venezuela,
and Brazil and has frequently been confused with S. incrustatum, which
it greatly resembles. It is generally associated with small to medium flowing
rivers in flat savanna areas. Shelley et al. (1997) described the morphological
differences and previous taxonomic confusion surrounding these two species.
In the present survey S. limbatum was collected from the southern
Rupununi District biting man and breeding on leaves in 1.5 m to 10 m wide
rivers. All the specimens previously identified by Davies (1973) as S.
incrustatum are S. limbatum. Preliminary studies on its man-biting
densities and insecticide trials with temephos have been reported (Humphrys
et al. 1977, Rambajan 1981 ab). Shelley et al. (1987) showed that this species
is capable of acting as a host to Onchocerca volvulus outside the
Amazonia onchocerciasis focus in Brazil and hence it may be a vector of M.
ozzardi in Guyana.
Simulium (Psaroniocompsa)
oyapockense Floch & Abonnenc (species complex)
This species was
originally described from French Guiana and is now known to be common and
widespread in South America. Shelley et al. (1997) reviewed its complicated
taxonomy and provided a full description of all stages. S. oyapockense s.l. is
a species complex in which most populations are anthropophilic and is closely
related to the anthropophilic S. roraimense Nunes de Mello. The two
species can only be separated morphologically in the male and pupa and hence
man-biting females of the two species cannot be distinguished. In neighbouring
Brazil both species occur, often sympatrically, and an atypical form of male S.
oyapockense s.l. occurs where the scutal cunae are extended into tails.
This atypical form also occurs in Guyana (Fig.
57), but no S. roraimense has yet been recorded. Most of the specimens
from Guyana were collected biting man and have been recorded as S. oyapockense
s.l. However, it is likely that S. roraimense will be found in
this country and at such a time the status of these females will need to
be re-examined. S. oya-pockense s.l. was commonly found in the Rupununi
District of Guyana biting man (see Material Examined).
Its breeding grounds are often difficult to locate and tend to be in rapids
in larger, fast running rivers. Humphrys et al. (1977) and Rambajan (1981b)
made observations on the biting habits of this species around the town of
Lethem in the southern Rupununi District (Map). S.
oyapockense s.l. is medically important because of its role as a vector
of two filarial species that infect man. In the Rupununi District and in
the adjacent savanna region of Brazil it is a vector of M. ozzardi (Nathan
et al. 1982, Moraes et al. 1985) and in the Amazonia onchocerciasis focus
in Brazil it is the primary vector of O. volvulus (Shelley et al.
1997).
Simulium (Psaroniocompsa)
quadrifidum Lutz
This zoophilic
species has been covered in Shelley et al. (1997) and is found in Bolivia,
Brazil, Ecuador, Surinam, and Venezuela. S. quadrifidum was collected
attached to dead leaves in small rapids, in 10 m wide rivers in the southern
part of the Rupununi District.
Simulium (Psilopelmia)
kabanayense Ramírez Pérez & Vulcano, 1973
Only three pupal
exuviae of this species exist in the BMNH collection. They were originally
identified as S. lutzianum (Smart 1940), but have now been identified
as S. kabanayense, a presumably zoophilic species only previously
recorded from Venezuela (Ramírez Pérez & Vulcano 1973,
Ramírez Pérez 1983, Hamada & Grillet 2001). The specimens
from Guyana were collected from a small stream in northern Rupununi District.
Simulium (Psilopelmia)
lutzianum Pinto, 1932
(species complex)
The species is
dealt with in Sawyer (1991) and Shelley et al. (1989a, 1997) and occurs in
Argentina, Bolivia, Brazil, Colombia, Ecuador, Peru, and Venezuela. It is
usually zoophilic, but has been recorded biting man in Southern Brazil (Shelley
et al. 2000). In Guyana pupae were collected on vegetation in small rapids
in 15 m wide rivers.
Simulium (Psilopelmia)
maroniense Floch & Abonnenc, 1946 (species complex)
This is a species
found in the R Orinoco tributaries of Southern Venezuela, the northern tributaries
of the R Amazon in Brazil and in the Guianas. There have been some taxonomic
problems concerning this species and the closely related S. rorotaense described
by Floch and Abonnenc (1946) in the same paper. In a paper reviewing the
taxonomy of several Neotropical species Shelley et al. (1984) regarded S.
maroniense s.l, S. wuayaraka Ortiz, 1957, S. fulvinotum Cerqueira & Nunes
de Mello in Cerqueira, 1967 and S. ignacioi Ramírez
Pérez and Vulcano, 1973 as synonyms of S. rorotaense.
During the publication of this paper Ramírez Pérez (1983) was
issued in which S. maroniense s.l. and S. rorotaense were treated
as valid species, S. wuayaraka was considered a synonym of S. maroniense
s.l. and S. ignacioi a synonym of S. ro-rotaense. A
discussion of these actions and subsequent treatment of these names by other
reviewers is given in Shelley et al. (1997), who maintained their original
synonymy list under S. rorotaense as expounded in their 1984 paper.
Since then Hamada and collaborators have carried out more extensive morphological
and cytological work and concluded that both S. maroniense s.l. and S.
rorotaense are valid species. Hamada and Adler (1998) were unable to
find differences between adult S. maroniense s.l. and S.
rorotaense, but in specimens collected from the Brazilian Amazon were
able to distinguish pupae in the following ways. S. maroniense s.l. has
thicker, more darkly pigmented gills, dorsal filaments shorter than ventral
and more basal branching. The filaments form a rosette in S. maroniense
s.l., but are bunched forwards in S. rorotaense. Trichome branching
and tubercle density in pupae were variable in each species [as we also found],
but generally tubercles were larger and more densely distributed in S.
maroniense s.l. In this paper they considered S. ignacioi to be
a valid species, but provided no evidence to support this view and apparently
did not examine type material. In their key the pupa is recorded as indistinguishable
from that of S. rorotaense, with which Ramírez Pérez
(1983) had synonymised it, based on the original description of S. ignacioi by
Ramírez Pérez and Vulcano (1973). A later paper by Hamada and
Grillet (2001) with keys to species in two adjacent regions of Southern Venezuela
and Northern Brazil stated that Hamada and Adler (1998) had based their recognition
of species status for S. ignacioi on gill filament numbers and chromosomal
configuration [although not stated in the paper]. In their key to last instar
larvae, Hamada and Grillet (2001) recorded the gill histoblast of S. ignacioi with
14-17 filaments (usually 16) [the original description of Ramírez
Pérez and Vulcano (1973) recorded 17-20 filaments in 8 specimens examined]
compared to S. maroniense s.l. and S. rorotaense with 17-23
filaments (usually 18-21). The pupa of S. ignacioi is again keyed
out with that of S. rorotaense, having 17-23 thin, lightly pigmented
filaments, not arranged in the form of a rosette [In couplet 17 of the key
an error exists where specimens with gills with "10-17 filaments" are
compared to those with gills "with more than 17 filaments"]. In
the key to pupae they also recorded S. ignacioi and S. rorotaense as
having lateral fenestrations in the cocoon, that are absent in S. maroniense
s.l.. We examined pupae of S. maroniense s.l. collected and identified
by Hamada and Xavier from Brazil and S. rorotaense collected by Hamada
and Fouque from French Guiana (see Material Examined) and both species showed
lateral fenestrations in the cocoon. Hamada and Adler (1999) provided cytological
evidence supporting the validity of S. maroniense s.l., which was
recorded as having four cytotypes in Brazil compared to the single cytotype
of S. rorotaense. The two species were separated by the presence of
the fixed terminal inversion IIIL-5 in S. maroniense s.l. Chromosomal
evidence was also used to support the synonymy of S. fulvinotum (Py-Daniel
1982, Shelley et al. 1984, 1997) with S. rorotaense, but insufficient
material was available for comment on the status of S. ignacioi and S.
wuayaraka. Subsequently, Hamada and Fouque (2001) showed a similar karyotype
for S. rorotaense topotypes from French Guiana, but were unable to
obtain S. maroniense s.l. from its type locality because of the construction
of a dam in the area. Scarpassa and Hamada (2003) were unable to find diagnostic
loci between S. rorotaense and a cytotype of S. maroniense s.l. in
recent studies on isoenzyme variation of several species from the Brazilian
Amazon.
We have now re-examined
the type specimens of S. maroniense s.l. and S. rorotaense, examined
named specimens of the two species loaned by Dr Neusa Hamada from the Inpa
collection, specimens from the BMNH collection, kindly reviewed by Dr Neusa
Hamada, and from CUAC. We have the following comments. We were only able
to locate the lectotypes and paralectotypes of the two species in the IP
in Paris. The other paralectotypes referred to in the original publication
and assumed by Shelley et al (1984) to be in the IP, French Guiana could
not be located there and are presumed lost (N Hamada, pers. commun.). The
difficulty in establishing whether S. maroniense s.l. is a synonym
of S. rorotaense still lies in the fact that the slide of the reared
female lectotype of the former species lacks the pupal exuviae and of the
male paralectotype all but the abdomen of the pupal exuviae, the stage in
which the only significant interspecific characters occur. Recourse has therefore
had to be made to the authors' original descriptions and subsequent work
by Hamada and collaborators. The original authors (Floch & Abonnenc,
1946) described S. maroniense s.l. as a species distinct from S.
rorotaense based on differences between the female genitalia and cibarium
and between pupae. Later, Coscarón (1990) distinguished the two species
based on the size of the sensory organ in the maxillary palp, the size of
the nudiocular triangle and minor differences in pupal morphology, all of
which are intraspecific variations. Our re-examination of the adult types
and specimens recorded in "Material Examined" revealed no differences
in adult morphology. In the pupae the original authors referred to the presence
of the following characters in S. rorotaense with observations for S.
maroniense s.l. in brackets - 2-3 branched trichomes (3-6), largely unpigmented
thorax and gill (finely pigmented), small and sparsely distributed tubercles
(strong and densely distributed) and no great difference in gill filament
lengths (shorter dorsally) and some secondary gill branches not basal (relatively
basal). We recorded the following for the female lectotype of S. rorotaense-
the thoracic trichomes vary from single to at least 5 branched (trichome
broken) and of the head bifid to 4-branched, no pigmentation is evident and
tubercles are relatively small and sparsely distributed, gill filaments are
approximately of equal length and secondary branching can occur up to the
middle part of the gill. Pupae in the BMNH collection and loaned from the
Inpa collection (see Material Examined) for S. maroniense s.l. showed
thoracic trichomes with 1-9 branches, pigmented thorax and gills, well developed,
densely distributed thoracic tubercles, dorsal gill filaments shorter than
ventral and branching always basal. The principal character of the rosette
form of the S. maroniense s.l. gill and its shorter dorsal filaments,
both not present in S. rorotaense, was efficient in separating most
specimens. However, in some (even preserved in spirit) it was difficult to
make an accurate identification, which might suggest the presence of a cline
with the rosette and forward bunched forms of the filaments representing
either extreme. Consequently, we agree with the re-validation of S. maroniense
s.l., but advocate an examination of larger series of specimens to assess
the variability of the form of the gill in different localities.
Our recognition
of the type status of S. maroniense s.l. now opens the question of
which names are synonymous with which species. It is now known from cytological
evidence that S. fulvinotum is a synonym of S. rorotaense (Hamada & Adler,
1999). We previously regarded S. ignacioi and S. wuayaraka as
synonyms of S. rorotaense (Shelley et al. 1984, 1997), while Ramírez
Pérez (1983) synonymised S. ignacioi with S. rorotaense and S.
wuayaraka with S. maroniense s.l. The types of S. ignacioi and S.
wuayaraka are now lost to science [The private collection of Dr Ramírez
Pérez is now in such a poor condition as to be of limited value (E
Grillet, pers. commun.) and is not freely accessible; the private collection
of Dr Vulcano is not available for examination and the other depositories
cited in the original descriptions have lost the specimens]. Therefore, we
can only consider the original descriptions of the two species concerned,
taking into account the comments made in Shelley et al. (1997) in relation
to subsequent publications by Ramírez Pérez on S. maroniense
s.l. and S. rorotaense. In addition to these we agree that the
figure of the gill of S. maroniense s.l. in Ramírez Pérez
(1983) is of this species because of the shorter dorsal filaments and the
gill of S. rorotaense appears to be of this species. The recent work
by Hamada and Grillet (2001) revealed only S. maroniense s.l. in
several watercourses in the Gran Sabana area of Venezuela and no S. rorotaense,
but higher sample numbers are required before any conclusions on distribution
patterns may be drawn. S. ignacioi is difficult to place because figures
of the original description (Ramírez Pérez & Vulcano 1973)
are different for this species in Ramírez Pérez et al. (1983),
which also has a more complete description. In the latter publication the
male scutal pattern, gill, female and male genitalia have all been redrawn
and these figures are replicated to illustrate S. rorotaense (with
which S. ignacioi is synonymised) in Ramírez Pérez (1983).
The latter paper figures the male scutum with 1+1 sub-median, comma-shaped
marks on the anterior scutal border, but no mention of these is made in the
text. We have now examined further material of S. maroniense s.l. from
the R Mucajai in Brazil and of S. rorotaense from the Reserva Ducke,
near Manaus in Brazil and have noted the presence of these marks in one male
of each species. They are of a lighter orange than the rest of the scutum
and are only visible when the light is anterior to the specimen with the
thorax tilted to an almost lateral position. We still regard S. ignacioi as
a synonym of S. rorotaense because the figure of Ramírez Pérez
and Vulcano (1973) shows secondary branching of the gill filaments less basal
than in S. maroniense s.l. and no evidence of the dorsal filaments
being shorter than the ventral, two of the main features typical of S.
rorotaense. S. wuayaraka was described from 16 man-biting females,
whose description is similar to those of S. maroniense s.l. and S.
rorotaense, and so in the absence of pupae no objective decision may
be made on synonymy. The distribution of S. maroniense s.l. and S.
rorotaense is not sufficiently known to use this as a basis for synonymy.
Therefore, for taxonomic simplicity we accept the synonym of S. wuayaraka with S.
maroniense s.l. recorded by Ramírez Pérez (1983).
Distribution records
for S. maroniense s.l. and S. rorotaense recorded in Shelley
et al. (1997) are accordingly amended as shown in the "Material Examined".
Those for Ramírez Pérez (1983) need to be verified.
Coscarón
(1990) placed S. maroniense s.l. and S. rorotaense in the subgenus Ectemnaspis.
Simulium (Psilopelmia)
perflavum Roubaud, 1906
Morphological descriptions
and taxonomic details of this species may be found in Coscarón (1990,
1991) and Ramírez Pérez (1983). Rambajan (1979) described S.
nilesi from the southern Rupunini District, but this name was later synonymised
with S. perflavum by Py-Daniel (1989). The zoophilic species occurs
in Argentina, Brazil, Paraguay, and Venezuela (Crosskey & Howard 1997).
In Guyana S. perflavum was collected in muddy, slow flowing rivers
of 1-5m wide with pupae attached to leaves and submerged grasses.
Simulium (Trichodagmia)
guianense Wise, 1911
(species complex)
This anthropophilic
species, which is a primary vector of human onchocerciasis in Brazil and
Venezuela, has been revised by Shelley et al. (1997). Since then four pinned
females have been found in the Nearctic accessions of the BMNH. These are
the four missing specimens of the 11 sent by Melville to Wise (Wise 1911).
These were presented by Dr KS Wise and bear printed labels, but not labels
in the hand of Wise (see Material examined for details). Extra labels have
been added to the lectotype and original paralectotypes indicating Melville
as collector (Wise had used his own name on the labels) and extra details
have been added to the four paralectotypes recently found in the BMNH. All
four specimens are S. guianense s.l. and the genitalia of two confirmed
this identification. They are undoubtedly paralectotypes and have been labelled
accordingly. This species is also found in Brazil, the other Guianas and
Venezuela. S. guianense s.l. was collected on submerged vegetation,
especially of the "Orin Weed" of the family Podostemaceae, in fast
flowing stretches close to waterfalls or steep rapids of 50 m wide rivers
with rocky beds.
Simulium (Trichodagmia) perplexum Shelley,
Maia-Herzog, Luna Dias & Couch, 1989
This anthropophilic
species is only known from its type locality at the Kaieteur Falls, Potaro
R. Pupae of S. perplexum were collected attached to submerged vegetation
in the family Podostemaceae by Smart (1940), who identified it as S. guianense
s.l. to which it is closely related. A full description of this species
is found in Shelley et al. (1989b).
MATERIAL
EXAMINED
Simulium (Chirostilbia)
spinibranchium Lutz, 1910
GUYANA
PINNED
Rupununi, Orinduik, ADC's Creek,
[site 32, (GUY 79/ 1c, GUY 79/2, GUY 74/4)], 4º43'N 60º02.9'W,
450 m; 3.xii.1970, (JB Davies) - 3 females, 1 male (reared, 1 without
pupa) (BMNH, B.M. 1999-16, specimen nos. 151227, 151228 151231). Rupununi,
Orinduik, ADC's Creek, [site 32, (GUY 70/1a)], 4º43'N 60º02.9'W,
450 m; 3.xii.1970, (JB Davies) - 1 male (reared) (BMNH, B.M. 1999-16,
specimen no. 151226).
SPIRIT
Rupununi, Orinduik, ADC's Creek,
[site 32, (GUY 70)], 4º43'N60º02.9'W; 2.xii, 1970, (JB Davies)
- 1 pupa (BMNH, B.M.1999-16, sample no. 151158). Rupununi, Orinduik, ADC's
Creek, (site 32, [GUY 70/6]), 4º43'N 60º02.9'W, 450 m; 2.xii.1970,
(JB Davies) - several pupae (BMNH, B.M.1999-16, sample no. 151165).
Simulium (Chirostilbia) subpallidum Lutz,
1910
GUYANA
PINNED
Rupununi, Orinduik, R. Ireng,
[site 13, (GUY 22/1; 23/2, 4; 25/1)], 4º44'N60º02.9'W, 450 m; 23.iii.1970,
(JB Davies) - 2 females, 2 males (reared) (BMNH, B.M.1999-16, specimen
no.151212, 151215, 151224, 151243). Rupununi, Orinduik, R. Ireng, (site 10,
[GUY 16/20]), 4º44.5'N 60º02.4'W, 460 m; 23.iii.1970, (JB Davies)
- 1 male (reared) (BMNH, B.M.1999-16, specimen no. 151210). Rupununi, Orinduik,
R Ireng, [site 11, (GUY 19/3)], 4º44.3'N 60º02.3'W, 460 m; 22.iii.1970,
(JB Davies) - 1 male (reared) (BMNH, B.M.1999-16, specimen no.151211).
Rupununi, Orinduik, R Ireng, (GUY 23/1), 4º44'N 60º02'W, 450 m;
23.iii.1970, (J.B. Davies) - 1 male (BMNH, no. 141214). Rupununi,
Orinduik, un-named stream (site 12, GUY 64/6; 68/2; GUY 66/1), 4º44.5'N
60º02'W, 455-460 m; 1.xii.1970, (JB Davies) - 2 males (reared,
but not associated with pupa) (BMNH, B.M.1996-16, no. 141218; 151259). Rupununi,
Orinduik, near R Ireng crossing, un-named stream [site 30, (GUY 64/3-48-9)],
4º44.5'N60º02.8'W, 460 m; 23.iii.1970, (JB Davies) - 4 females,
1 male (reared, two but not associated with pupae) (BMNH, B.M.1999-16, sample
no.151216-21). Rupununi, Orinduik, ADC's Creek, [site 32, (GUY 70/2)], 4º43'N
60º02.9'W; 2.xii. 1970, (JB Davies) - 1 female (reared) (BMNH,
B.M.1999-16, specimen no. 151225). Rupununi, Orinduik, R Tumong, (site 33,
[GUY 70]), 4º42.8'N 60º02.4'W; 2.xii.1970, (JB Davies) -
1 female (reared, but not associated with pupa) (BMNH, B.M.1999-16, specimen
no. 1511223). Rupununi, Orinduik, R Chiung, [site 41, (GUY 80/3a, 4)], 4º39.3'N
59º50.5'W; 5.xii.1970, (JB Davies) - 1 female, 1 male, (reared,
but not associated with pupae) (BMNH, B.M.1999-16, specimen no. 151222).
Rupununi, Orinduik, R Chiung, [site 46, (GUY 80/3a; 88/7-8)], 4º39.2'N59º50.5'W;
7.xii.1970, (JB Davies) - 4 females, 2 males, (reared, but not associated
with pupa) (BMNH, B.M.1999-16, specimens no. 151238, 151282-86). R Kumu,
[site 58, (GUY 105)], 3º19'N 59º46.5'W, 143m; 17.ii.1975, (JB
Davies) - 1 male (reared, but not associated with pupa) (BMNH, B.M.1999-16,
specimen no.151271).
Simulium (Inaequalium)
clarki Fairchild, 1940
GUYANA
PINNED
R Tumong, 3.xii.1970, (JB Davies)
- 4 females (man-biting) (BMNH). Orinduik, Sagars' house, 4º44'N60º2'W;
28.vii.1969, (Sagar) - 19 females (man-biting) (BMNH). Creek near
District Commissioner's house, 4º42'N60º1'W; 3.xii.1970, (JB
Davies) - 15 females (man-biting) (BMNH). Orinduik, R Ireng, 4º44'N60º2'W;
23.iii.1970, (JB Davies) 1 female (man-biting) (BMNH). Kato, R.Chiung,
4º44'N60º2'W; 5.xii, 7.xii.1970, (JB Davies) - 14 females
(man-biting) (BMNH). Kurubakaru, R. Tumong, 4º38'N59º52'W; 2.xii.1970,
(JB Davies) - 5 females (man-biting) (BMNH). R Tumong, between Habec
crossing and Santa Maria; 2.xii.1970, (JB Davies) - 1 female (man-biting)
(BMNH). R Tumong, lower falls; 2.xii.1970, (JB Davies) - 1 female
(man-biting) (BMNH). Kato, water falls behind school, 4º40'N59º41'W;
10.iii.1970, (JB Davies) - 12 females (man-biting) (BMNH). Orinduik,
R. Ireng; 11.vii.1957, (R.McConnell) - 4 females (BMNH). Rupununi,
Kato, R Chiung, [site 46, (GUY 89/1-2, 4)], 4º39.3'N59º50.5'W,
460 m; 7.xii.1970, (JB Davies) - 3 females (man-biting) (BMNH, specimens
no. 151291-92, 94). Rupununi, Kato, R Chiung, [site 48, (GUY 91/2)], 4º38.9'N59º49.6'W,
460 m; 8.xii.1970, (JB Davies) - 1 female (man-biting) (BMNH, specimens
no. 151287). Rupununi, Kato, R Ireng, [site 11, (GUY 17/4)], 4º44.3'N60º02.3'W,
460 m; 21.iii.1970, (JB Davies) - 1 female (man-biting) (BMNH, specimens
no. 151296).
SLIDES
Kato, R Chiung, 4º44'N60º2'W;
5.xii, 7.xii.1970, (JB Davies) - 2 females (man-biting) (BMNH). Creek
near District, Commissioner's house, 4º42'N60º1'W; 3.xii.1970,
(JB Davies) - 1 female (man-biting) (BMNH). Kato, tributary of R Kowa;
6.xii.1970, (JB Davies) - 1 female (man-biting) (BMNH). R Tumong,
between Habec crossing and Santa Maria; 2.xii.1970, (JB Davies) -
1 female (man-biting) (BMNH). R Tumong, Kurubakaru trail; 3.xii.1970, (JB
Davies) - 1 female (man-biting) (BMNH). Rupununi, Orinduik, R Ireng,
[site 1, (GUY 17/2)], 4º44.3'N60º02.3'W, 460 m; 21.iii.1970, (JB
Davies) - 1 female (reared but not associated with pupa) (BMNH, B.M.
1999-16, specimen no. 151290). Rupununi, Kato, R Chiung, [site 46, (GUY 89/3)],
4º39.3'N59º50.5'W, 460 m; 7.xii.1970, (JB Davies) - 1 female
(man-biting) (BMNH, specimen no. 151293). Rupununi, Orinduik, R Ireng, [site
15, (GUY 28/1)], 4º43.1'N60º03'W, 450 m; 23.iii.1970, (JB Davies)
- 1 female (man-biting) (BMNH, B.M. 1999-16, specimen no. 151288). Rupununi,
Orinduik, R Ireng, [site 11, (GUY 17/1)], 4º44.3'N60º02.3'W, 460
m; 21.xi.1970, (JB Davies) - 1 female (man-biting) (BMNH, B.M. 1999-16,
specimen no. 1511289). Orinduik, R Tumong; 12.xii.1970, (JB Davies)
- 3 females (man-biting) (BMNH). Rupununi, Kato, un-named river, [site 49,
(GUY 92)], 4º40.3'N459º42.9'W; 10.xii.1970, (JB Davies)
-1 female (man-biting) (BMNH, BMNH 1999-16, specimen no. 151178).
SPIRIT
Orinduik, R Tumong; 12.xii.1970,
(JB Davies) - 12 females (man-biting) (BMNH).
Simulium (Notolepria) exiguum Roubaud,
1906 (species complex)
BRAZIL
State of Roraima
PINNED
Rio Tacutu, (site
1315); 28.xi.1994, (S Luz & APA Luna Dias) - 1 female,
2 males (reared) (BMNH).
Simulium (Psaroniocompsa) cauchense Floch & Abonnenc,
1946
GUYANA
PINNED
Rupununi, Orinduik, R Ireng, [site
10, (GUY 16/13,14, 16a, 17)], 4º44.5'N60º02.4'W, 460 m; 21.iii.1970,
(JB Davies) - 6 females, 1male (reared) (BMNH, B.M. 1996-16, specimen
nos. 151180, 151182, 151200, 151201, 151181-83). Rupununi, Orinduik; 22.iii.1970, (JB
Davies) - 1female (BMNH). Rupununi, Orinduik, R Ireng, [site 11, (GUY
19/1,6,8)], 4º44.3'N60º02.3'W, 460 m; 22.iii.1970, (JB Davies)
- 2 males (reared) (BMNH, B.M. 1996-16, specimens no. 151185-86, 88). Rupununi,
Orinduik, R Tumong, [site 31, (GUY 65/1)], 4º42.4'N60'02.8'W, 450 m;
1.xii.1970, (JB Davies) 1 male (reared but associated with pupae)
(BMNH, B.M. 1996-16, specimen no. 151206). Rupununi, Orinduik, R Tumong,
[site 33, (GUY 72/6)], 4º42.8'N60'02.4'W, 450 m; 2.xii.1970, (JB
Davies) - 1 male (reared but not associated pupa) (BMNH, B.M. 1996-16,
specimen no. 151190). Creek on route to R Kowa, un-named river, [site 43,
(GUY 83/1)], 4º38.3'N59º48.8'W, 460 m; 6.xii.1970, (JB Davies)
- 1 male (reared but without pupa) (BMNH, B.M. 1996-16, specimen no151260).
Rupununi, Kato, R Chiung, [site 41, (GUY 80/2a; 83/2)], 4º30.3'N59º50.5'W,
460 m; 3.xii.1970, (JB Davies) - 2 males (reared, but not associated
with pupae) (BMNH, B.M. 1996-16, specimens no 151209, 151261). Rupununi,
Lethem, R Mocomoco, [site 3, (GUY 102)], 3º21.3'N 49'47.4'W, 140 m;
10.i.1970, ([JB Davies) - 1 male (reared) (BMNH, B.M. 1996-16, specimen
no. 151263) Rupununi, Lethem, R Burru, [site 3, (GUY 102)], 3º17.6'N59'49.3'W,
145m; 13.i.1975, (JB Davies) - 1 male (reared) (BMNH, B.M. 1996-16,
specimen no. 151269).
Simulium (Psaroniocompsa)
limbatum Knab, 1915
TYPE MATERIAL
GUYANA
PINNED
Rupununi District, R Rupununi; ix.1913,
(KS Wise) - 1 female [HOLOTYPE], 5 females [PARATYPES] (BMNH).
OTHER MATERIAL
PINNED
Rupununi, Orinduik, R.D.C's, [site
32, (GUY 79/1, 1b)], 4º44'N 60º02'W, 450 m; 3.xii.1970, (JB
Davies) - 2 males (BMNH, no. 151229-30). Rupununi, Lethem, R Tabatinga,
[site 22, (GUY 45/1-2)], 3º22.5'N59º47.6'W, 145m; 27.xii.1970,
(JB Davies) - 2 females (man-biting) (BMNH, B.M. 1996-16, specimen
nos. 151298-99). Rupununi, Le-them, R Mocomoco, [site 21, (GUY 43/1)], 3º20.9'N
59º46.8'W, 145m; 7.xii.1970, (JB Davies) - 1 female (man-biting)
(BMNH, B.M. 1996-16, specimen nos. 151272). Rupununi, Lethem, un-named creek,
[site 27, (GUY 55/1)], 3º23.5'N 59º46'W, 144m; 28.xi.1970, (JB
Davies) - 1 female (reared but not associated with pupa) (BMNH, B.M.
1996-16, specimen no. 151323). Rupununi, Lethem, R Burru, [site 8, (GUY 51/1-10)],
3º17.6'N 59º49.3'W, 145m; 28.xi.1970, (JB Davies) - 4 females,
6 males (reared but not associated with pupae) (BMNH, B.M. 1996-16, specimen
nos. 151304-13). Manari; 14.vii, 1975, (E.S.Tikasingh) - 2 females
(BMNH). Rupununi Lethem, R Kumu, [site 7, (GUY 49/4, 6)], 3º18.3'N 59º48.8'W,
145m; 28.xi.1970, (JB Davies) - 2 males (reared but not associated
with pupae) (BMNH, B.M. 1996-16, 151274, 151276). St Ignatius, R. Mocomoco,
(site 14); 20.iii.1970, 10.i.1975, (JB Davies) - 11 females (man-biting),
2 males (reared) (BMNH). Bridge at R. Mocomoco; 11.i.1975, (JB Davies).
Lethem, 1 mile above St Ignatius crossing, R Mocomoco, 27.xi.1970, (JB
Davies) - 3 females, 2 males (reared) (BMNH). Cen. Ranch, R Kuma; 17.i.1975,
(JB Davies) - 4 females (reared) (BMNH). Francis' Ranch, R. Kuma;
17.i.1975, (JB Davies) 1 female (reared) (BMNH). Bridge at R Kuma
(sites 11 & 12); 20.iii.1970, 28.xi.1970, (JB Davies) - 14 females
(man-biting), 2 males (reared) (BMNH). R Burru, at crossing, (site 13); 20.iii.1970,
(JB Davies) - 2 females (man-biting) (BMNH). MEP (Malaria Eradication
Programme) Camp, R Kumu; 28.xi.1970, (JB Davies) - 40 females (man-biting)
(BMNH). R Burru; 13.i.1970, (no collector's name)-2males (reared) (BMHH).
Near Lethem, R Burru, at crossing, 3º17'N59º49'W; 28.xi.1970, (JB
Davies) - 10 females (man-biting). Near Lethem, confluence of R Ireng
with R Tabatinga, 3º23'N59º47'W; 27.xi.1970, (JB Davies)
- 10 females (man-biting). Near Lethem, R Tabatinga, 3º21'N59º48'W;
27.xi.1970, (JB Davies) - 17 females (man-biting) (BMNH). Near Lethem,
Manari Creek; 16.vi.1977, (A.J.Shelley) - 5 females (man-biting) (BMNH, B.M.
1979-580).
SPIRIT
Rupununi District, bridge at R
Kumu; 20.iii.1970, 28.xi.1970, (JB Davies) 1 female (man-biting),
1 female, 1 male (reared) (BMNH).
Simulium (Psaroniocompsa) oyapockense Floch & Abonnenc,
1946 (species complex)
GUYANA
PINNED
British Guiana (no locality, but
probably Rupununi or Siparuni rivers); 1908, (KS Wise on label probably Melville)
- 5 females (man-biting) (BMNH, B.M. 1908-207). Rupununi, Orinduik, R Ireng,
[site 15, (GUY 28/2)], 4º43.1'N60º03'W, 450 m; 23.iii.1970, (JB
Davies) - 1 female (man-biting) (BMNH, B.M. 1999-16, specimen no. 151297).
Itanimi Creek; 1908, (KS Wise) - 11 females (BMNH). British Guiana;
(no date), (Wise) - 2 females (BMNH, B.M. 1939-586). Tukeit; 31.viii.1937,
(Richards & Smart) - 3 females (man-biting) (BMNH, B.M. 1937-776).
Warratuk; 31.viii.1937, (Richards & Smart) - 6 females (man-biting)
(BMNH, B.M. 1937-776). Kaieteur savannah; 1, 4. & 6.i.1937, (Richards & Smart)
- 3 females (man-biting) (BMNH, B.M. 1937-776). Orinduik Falls, R Ireng;
2.viii.1957, (R. McConnell) - 2 females (BMNH). Orinduik, R Ireng,
4º44'N60º2'W; 23.iii.1970, (no collector's name)- 105 females (man-biting)
(BMNH). Rupununi District, Orinduik, R Ireng, 4º44'N60º2'W; 23.iii.1970,
(JB Davies) - 12 females (man-biting) (BMNH). Orinduik, near ADC's
house, 4º42'N60º1'W; 3.xii.1970, (JB Davies) - 41 females
(man-biting) (BMNH). Rupununi District, Orinduik, below lower fall; 24.iii.1970,
(JB Davies) - 3 females (man-biting) (BMNH). Rupununi District, Orinduik,
above and below falls; 24.iii.1970, (JB Davies) - 7 females (man-biting)
(BMNH). Rupununi District, R Tumong; 24.iii.1970, (JB Davies) - 3
females (man-biting) (BMNH). Rupununi District, Kurukabaru Trail, R Tumong,
4º47'N59º58'W; 3.xii.1970, (JB Davies) - 22 females (man-biting)
(BMNH). Rupununi District, between Habec crossing and Santa Maria, R Tumong;
2 & 3.xii.1970, (JB Davies) - 15 females (man-biting) (BMNH).
Rupununi District, Lower Falls, R Tumong; 2.xii.1970, (JB Davies)
- 17 females (man-biting) (BMNH). Rupununi District, Kato, R Chiung, 4º38'N59º52'W;
5 & 7.xii.1970, (JB Davies) 17 females (man-biting) (BMNH). Rupununi,
Apoteri, R Essequibo, ix-x.1926, (LD Cleare) - 20 females (man-biting)
(BMNH). Rupunini, Karanambu; viii.1959, (E McTurk) - 4 females (on
same pin, man-biting) (BMNH). British Guiana; (no date), (KS Wise)
- 4 females (BMNH). British Guiana, (no. 1611); ix.1913, (KS Wise)
- 10 females (man-biting) (BMNH, B.M. 1990-107, ex. Wellcome Coll.). Lethem;
15.viii.1975, (ES Tikasingh). Lethem Hospital, R Takatu; 21.vi.1977,
(AJ Shelley) - 7 females (man-biting) (BMNH, B.M. 1979-580). Lethem,
rest house, R Takatu; 14.vi.1977, (AJ Shelley) - 6 females (man-biting)
(BMNH, B.M. 1979-580). Rupununi District, Dadanawa, R Rupununi; 19.vi.1977,
(AJ Shelley) - 2 females (man-biting) (BMNH, B.M. 1979-580). Rupununi
District, Lethem, Bon Fim, R Takatu; 13.vi.1977, (AJ Shelley) - 2
females, 2 males (reared, pupa on slides) (BMNH, B.M. 1979-580).
SLIDES
Rupununi District, Lethem, Bon
Fim, R Takatu; 13.vi.1977, (AJ Shelley) - 2 females, 2 males (pupa,
adult pinned) (BMNH, B.M. 1979-580).
SPIRIT
Orinduik, R Ireng, (coll. No.
21/1); 22.iii.1970, (JB Davies) - several females (man-biting) (BMNH).
Orinduik, R Ireng, 2.iii.1969, (JB Davies) - several females (man-biting)
(BMNH). Orinduik, creek behind house of ADC; 22.iii.1969, 24.iii.1970, (JB
Davies) - several females (man-biting) (BMNH). Orinduik, beach, below
falls, R Ireng, (coll. 28); 2.iii.1969, (JB Davies) - 4 females (man-biting)
(BMNH). (No locality); 19.ix.1980, (M Nathan) - 2 females (man-biting)
(BMNH). Rupunini, Karanambu; viii.1959, (E McTurk) - several females
(man-biting) (BMNH). Takatu River; 4.21.1970, (no collector's name) - several
females (man-biting, from 4-6 pm) (BMNH). R Takatu, crossing at Bon Fim;
13.i.1975, (JB Davies) - 3 females (man-biting). Nr. Uiramutã,
R Ireng, (Brazilian side of river); 31.x.1997, (AJ Shelley & APA Luna
Dias) - numerous females (man biting) (BMNH). Lethem, behind government
house, (FR4-19); 15.viii.1975, (ES Tikasingh) - several females (man-biting)
(BMNH). Orinduik, (col. 126); 16.iv.1968, (J Darlington) - several
females (man-biting) (BMNH). (No locality given); 25.ix.1980, (M Nathan)
- 2 females (man-biting) (BMNH).
Simulium (Psaroniocompsa)
quadrifidum Lutz, 1917
GUYANA
PINNED
Rupununi, Lethem, R Mocomoco,
[site 26, (GUY 104)], 3º18.5'N59º43'W, 146 m; 13.i.1975, (JB
Davies) - 1 female (reared) (BMNH, B.M. 1999-16, specimen no. 1511270).
SPIRIT
Rupununi Lethem, R Mocomoco, [site
53, (GUY 10)], 3º18.8'N59º40'W, 144 m; 14.i.1970, (JB Davies)
- 1 male (reared) (BMNH, B.M. 1999-16, specimen no. 151127). Rupununi, Lethem,
R Tumong, [site 16, (GUY 32)], 4º42.9'N60º02.2'W, 450 m; 24.iii.1970,
(JB Davies) - 3 pupae (BMNH, B.M. 1999-16, sample no. 151148).
Simulium (Psilopelmia) kabanayense Ramírez
Pérez & Vulcano, 1973
GUYANA
SLIDES
(Originally identified as Simulium
lutzianus): Kaieteur savananah, small stream; 6.ix.1937, (S. Smart)
- 3 pupae (on same slide) (BMNH, B.M. 1937-778).
Simulium (Psilopelmia) lutzianum Pinto,
1932
(species complex)
GUYANA
PINNED
Rupununi, Orinduik, R Tumong,
[site 16, (GUY 30/3a; 77/3)], 4º42.9'N 60º02.2'W, 450 m; 24.iii.,
3.xii.1970, (JB Davies) - 2 females (reared, but not associated with
pupae) (BMNH, B.M. 1996-16, specimen no. 151213, 151236). Rupununi, Orinduik,
R Tumong, [site 33, (GUY 72/8)], 4º42.8'N 60º02.4'W, 450 m; 2.xii.1970,
(JB Davies) - 1 female (reared, but not associated with pupa) (BMNH,
B.M.. 1996-16, specimen no. 151237). Rupununi, Kato, R Kowa (Seyun), [site
44, (GUY 85/1-3, 5)], 4º69'N59º48'W, 400 m; 6.xii.1970, (JB
Davies) - 3 females, 1 male (reared but not associated with pupae) (BMNH,
B.M. 1996-16, specimen no. 151232-33, 151281). Kaieteur savannah; 4.ix.1987,
(no collector's name) - 1 male (at light trap) (BMNH).
Simulium (Psilopelmia) maroniense Floch & Abonnenc,
1946 (species complex)
Note: most specimens
from the BMNH collection recorded as S. rorotaense in Shelley et al.
(1997) are now placed as S. maroniense s.l.
TYPE MATERIAL
FRENCH GUIANA
SLIDES
Coeur Maroni; 12.viii.1945,
(no collector's name) - 1 female (reared, pupal pelt missing)
(LECTOTYPE, no. 709), 1 male (reared, pupal thorax and cocoon
missing) (PARALECTOTYPE, no. 708) (IP).
OTHER MATERIAL
BRAZIL
State of Amazonas
PINNED
Km 26 Estr. Manaus, Ducke Reserve,
Igarapé Acará; 20.i.1976, (Faustino) - 1 female (reared)
(BMNH).
State of Mato Grosso
PINNED
Dardanelos Falls,
R Aripuanã, 10°11'S 59°48'W; 22.iii.1977, (no
collector's name) - 2 man-biting females, (BMNH).
State of Pará
SPIRIT
Carajás District;
vi.1983 (L Ryan) - 1 female (man-biting) (BMNH).
State of Roraima
PINNED
Pacaraima, BR 174,
tributario do Igarapé Sargento Ávila, 04º 26'N
61º 07' W; 6.vi.1996, (N Hamada) - 2 females 2 males
(reared) (INPA). Mission post, R Auaris; 31.iii.1977, (RR.
Pinger) - 1 female (reared), 1 male (reared), (BMNH, B.M.1979-580).
Mission post, R Mucajaí, (site 285c); 6.i.1977, B.M.1979-580
(AJ Shelley & APA Luna Dias) - 1 female (reared, pupal
exuviae on slide) (BMNH). R Mucajaí, 200 m below Igarapé Coroconaí,
21.vii.1984 (AJ Shelley & APA Luna Dias) - 2 females
(reared), 2 males (reared, fore leg claw mounted), (BMNH). Near
mission post, R Mucajaí, Igarapé Coroconaí,
21.vii.1984 (AJ Shelley & APA Luna Dias), 1 male (reared)
(BMNH). R Preto, tributary of R Ajaraní; 28-29.iv.1979,
(R.W Crosskey & AJ Shelley) - 1 female (reared) (BMNH,
B.M.1979-258). Cachoeira, R Cauamé; 29.iv.1982 (APA
Luna Dias & R Malaguti) - 2 females (reared), 1 male
(reared) (BMNH). Boa Vista-Sta. Helena road, Igarapé Ávila;
29.xi.1980 (AJ Shelley & APA Luna Dias) - 1 female
(reared; fore leg claw mounted) (BMNH).
SLIDES
Pacaraima do Samã, (no.
88); 6.vi.1996, (N. Hamada) - 2 females, 1 male (reared) (Inpa). Pacaraima,
BR 174, Igarapé Sargento Alves, 04º26'N61º07'W; 14.i.1996,
(N Hamada) - several pupae (on three slides) (Inpa). Surucucus, Igarapé Falemu
(above hydroelectric dam), (site 791); 10.xii.1986 (AJ Shelley & APA
Luna Dias) 1 female (reared) (BMNH). Surucucus, Dalem; 11.xii.1986 (AJ
Shelley & APA Luna Dias) - 1 female (reared) (BMNH). Mission post,
R Mucajaí, 6.i.1977, (AJ Shelley) - 1 pupa (adult pinned) (BMNH,
B.M.1979-580). Near mission post, R Mucajai, Igarapé Coroconaí;
21.vii.1984 (AJ Shelley & APA Luna Dias) 1 female (reared), 1
pupa, 3 larvae (BMNH). R Mucajai, 200 below Igarapé Coroconaí,
(site 675-1); 21.vii.1984 (AJ Shelley & APA Luna Dias) 1 male
(reared) (BMNH). Mission post, R Auaris, 7.vii.1976, (AJ Shelley)
- 1 pupa, (BMNH, B.M.1979-580). Surucucus, Cachoeira 2 km from Funai Post,
(site 613); 7.v.1982, (APA Luna Dias & R. Malaguti) - 2 males
(reared) (BMNH). Surucucus, Igarapé Falemu (above hydroelectric dam),
(site 791); 10.xii.1986 (AJ Shelley & APA. Luna Dias) - 2 males
(reared) (BMNH). Uiramutã, Cachoeira do Urucá, (site 1303);
13.x.1997, (AJ Shelley & APA Luna Dias) - 1 male (reared) (BMNH).
SPIRIT
Surucucus, waterfall 2 km
from Funai post; 7.v.1982 (APA Luna Dias & R. Malaguti) 2 females
(reared), 7 males (reared), 3 pupae, (BMNH). Surucucus, Igarapé Falemu
(above hydroelectric dam); 10.xii.1986 (AJ Shelley & APA Luna Dias)
- 2 males (reared) (BMNH). Near mission post, R Mucajaí, Igarapé Coroconaí;
21.vii.1984 (AJ Shelley & APA Luna Dias) - 8 pupae, numerous larvae,
(BMNH). Mucajaí mission, Igarapé Coroconaí; 6.i.1977
(AJ Shelley) - 1 pupa, (BMNH). Mucajaí, 200 m below Igarapé Coroconaí,
21.vii.1984 (AJ Shelley & APA Luna Dias) 1 male (reared), 9 pupae,
18 larvae, (BMNH). Boa Vista-Sta. Helena road, Boca da Mato, Igarapé Cunaen;
11.viii.1984 (AJ Shelley & APA Luna Dias) - 1 male, 3 pupae, 6
pupal exuviae, (BMNH). Boa Vista-Sta. Helena road, Boca da Mato, Igarapé Cunaen,
small stream; 11.viii.1984 (AJ Shelley & APA Luna Dias) 3 females
(mass reared), 1 pupa, (BMNH). Auaris mission post, Igarapé; 9.vii.1979,
(AJ Shelley) - 1 pupa, (BMNH, B.M.1979-580). Mission post, R Auaris;
7.vii.1976, (AJ Shelley) & 8.xii.1986 (AJ Shelley & APA
Luna Dias) - 1 male (reared), 1 pupal exuviae, 3 larvae, (BMNH, B.M.1979-580).
Tributário do Ig. Samã, antes da casa da D. Alade, (site 88);
6.vi.1996, (N Hamada.) - several pupae (INPA). Igarapé do Banana;
04º25'N 61º13'W; 14.i.1996, (N Hamada) - several pupae (CUAC).
Igarapé Sargento Ávila; 04º26'N 61º07'W; vi.1996,
(N Hamada) - several pupae (CUAC).
GUYANA
PINNED
Rupununi, Lethem, R Mocomoco,
[site 6, (GUY 9/1-9)], 3º17.5'N59º37.8'W, 146 m; 19.iii.2970, (JB
Davies) 4 females, 4 males (reared, 1 male without associated pupa) (BMNH,
B.M. 1999-16, specimens nos. 151191-98). (Previously identified as S.
rubrithorax.) Kaieteur, Savannah; 6.ix.1937, (Richards & Smart)
- 1 female (at light trap) (BMNH).
SPIRIT
Rupununi, Lethem, R Nappi, [site
1, (GUY 1)], 3º21'N59º28'W, 146 m; 18.iii.1970, (JB Davies)
- 1 pupa (BMNH, B.M. 1999-16, sample no. 151101). Rupununi, Lethem, R Nappi,
[site 1, (GUY 3)], 3º21'N59º28'W, 146m; 18.iii.1970, (JB Davies)
- numerous pupae (BMNH, B.M. 1999-16, sample no. 151103). Rupununi, Lethem,
R Nappi, [site 2, (GUY 2)]; 18.iii.1970, (JB Davies) - 4 pupae (BMNH,
B.M. 1999-16, sample no. 151108). Rupununi, Lethem, R Mocomoco, [site 6,
(GUY 9/11)], 3º17.5'N59º37.8'W, 146 m; 19.iii.2970, (JB Davies)
- 1 female 3 males, numerous pupae (BMNH, B.M. 1999-16, sample no. 151108).
SLIDES
[Originally identified as S.
rubrithorax]: Kaieteur, savannah; 4.i.1937, (J Smart) - 1male
(only genitalia) (BMNH, B.M. 1937-778). [Originally identified as S. rubrithorax]:
Kaieteur, savannah; 4 ix.1937, B.M. (J Smart), 1 male (genitalia and
single hind leg only) (BMNH, 1937-778). [Originally identified as S. rubrithorax]:
High forest, sandstone bed of stream, Kaieteur/Tukeit Trail, 9.ix.1937, (J.
Smart) - 3 males (dissected from pupae) (BMNH, B.M.1937-778).
VENEZUELA
Amazonas Territory
PINNED
[Originally identified
as S. wuayaraka Ortiz]: Parima, 18.iv.1980, (no collector's
name) - 1 male (reared) (BMNH).
Bolivar State
PINNED
[Originally identified
as S. ignacioi Ramírez Pérez & Vulcano]:
Wonaven, (no date or collector stated) - 1 male (reared)
(BMNH).
Simulium (Psilopelmia)
rorotaense Floch and Abonnenc, 1946
TYPE MATERIAL
FRENCH GUIANA
SLIDES
Rorota; 31.v.1946,
(no collector's name)-1 female (reared) [LECTOTYPE, no.
751], 1 male (reared but without pupa) [PARALECTOTYPE, no. 752]
(IP).
OTHER MATERIAL
BRAZIL
State of Amazonas
PINNED
Manaus, Reserva Ducke,
Am 010-km 22, Igarapé Barro Branco, 2º55' S 59º 59'W;
18.xi.1998, (N. Hamada) - 1 female, 2 males (reared but
not associated with pupae) (Inpa). Manaus, Reserva Ducke, Inpa,
Ig. Acará, 2º57'S59º57'W; 11.vii.1995 (N.
Hamada) - 1 female (reared, head, wings, legs and genitalia
on slide, rest of body pinned) (BMNH), 1 female, 1 male (reared)
(CUAC). P Figueiredo, BR 174, Igarapé Lajes, Balneário,
(site 69), 01º59'S 60' 01º 29'W; 19.vii. 1996, (N.
Hamada) - 1 male (reared, genitalia and wings on slide) (BMNH).
Manaus-Itacoatiara road, Reserva Ducke, Ig. Acará; 20.i.1976,
(Faustino) - 1 female (reared) (BMNH.)
SLIDES
Manaus, Reserva Ducke, Am 020,
km 26, Igarapé Acará; 21.vi.1996, (N. Hamada) - several
pupae (Inpa). P Figueiredo, BR 174, Igarapé Lajes, Balneário,
(site 69), 01º59'S 60' 01º 29'W; 19.vii.1996, (N. Hamada)
- 1 male (reared) (genitalia and wings on slide, rest pinned) (BMNH). Presidente
Figueiredo, Am 240, pequeno Igarapé, acima da caverna da Maruaza,
02º02'S59º58'W; 22.vi.1996, (N Hamada) - several pupae (Inpa).
Manaus, Reserva Duke, 03º00'N59º56'W; 21.vi.1996, (N Hamada)
1 female, 1 male (mass reared), 2 pupae (BMNH), 1 female, 1 male (mass reared),
2 pupae (CUAC).
SPIRIT
Manaus, Reserva Duke, 03º00'N59º56'W;
21.vi.1996, (N Hamada) - 1 male, several pupae (BMNH), several females,
males, several pupae (CUAC).
State of Pará
SLIDES
Estrada Santarém,
Cuiabá, BR 163, 28 km do Ramal da Jahoti, 02º53'S54º45'W;
2.viii.1996, (N Hamada) - several pupae (Inpa).
FRENCH GUIANA
SPIRIT
Montagne du Mahery,
Rorota; 21.6.1999, (N Hamada & F Fourque) -
several pupae (Inpa).
Simulium maroniense
s.l./ Simulium rorotaense
Specimens under
this title are from localities where no reared material is available to provide
a more precise identification.
BRAZIL
State of Mato Grosso
PINNED
Dardanelos Falls,
Rio Aripuanã, (site 307), 10º11'S 59º48'W; 22.iii.1977,
(no collector name) - 2 females (man-biting) (BMNH).
State of Pará
SLIDE.
Carajas District,
vi.1983, (L Ryan), - 1 female (man-biting), (BMNH).
State of Roraima
PINNED
Arabobo Mt., (no.
3325); 24.xi.1952, (JG Myers) - 1 female (reared) (BMNH).
GUYANA
PINNED
Kutari Sources; i.ii.1936, (GA
Hudson) - 1 male (reared, but no associated with pupa) (BMNH, B.M. 1956-360).
Simulium (Psilopelmia)
perflavum Roubaud, 1906
GUYANA
PINNED
Rupununi, Lethem, R Burru, [site
8, (GUY 13/1-4)], 3º17.6'N59º49.3'W, 145 m; 20.iii.1970, (JB
Davies) - 1 female, 3 males (reared) (BMNH, B.M. 1999-16, specimens no.
151239, 151240, 151241, 151242). Rupununi, Lethem, R Burru, [site 8, (GUY
51/11-18)], 3º17.6'N59º49.3'W, 145m; 28.iii.1970, (JB Davies)
- 6 females, 2 males (reared, but not associated with pupa) (BMNH, B.M. 1999-16,
specimens no. 151314-151320). Rupununi, Lethem, small stream nr. St. Ignatius,
un-named river, [site 25, (GUY 52/1-10, 12)], 3º20.6'N59º47.8'W,
145m; 28.iii.1970, (JB Davies) - 6 females, 4 males (reared, but not
associated with pupa) (BMNH, B.M. 1999-16, specimens nos. 151322, 151328-151330,
151331-151336, 151338, 151242). Rupununi, Orinduik, Creek behind ADC's house,
un-named river, (site 12, (GUY 66/3-8; 68/1)), 4º43.8'N60º02'W,
455m; 1.xii.1970, (JB Davies) - 6 females, 1 male (reared, but not
associated with pupa) (BMNH, B.M. 1999-16, specimens no. 151157, 151251-52,
151254-57). Rupununi, R Tabatinga, (site 22, (GUY 66/3-8; 68/1)), 3º22.3'N59º47.6'W,
145m; 10.i.1975, (JB Davies) - 1 female (reared) (BMNH, B.M. 1999-16,
specimens no. 151262). Rupununi, R Burru, [site 8, (GUY 103/1)], 3º17.6'N59º47.3'W,
145m; 13.i.1975, (JB Davies) - 3 females, 2 males (reared) (BMNH,
B.M. 1999-16, specimens no. 151264-268).
SLIDES
Rupununi, Lethem, small stream
nr. St. Ignatius, unnamed river, [site 25, (GUY 52/6, 11, 13-14)], 3º20.6'N59º47.8'W;
28.iii.1970, (JB Davies) - 2 females, 2 males (reared, but not associated
with pupae) (BMNH, B.M. 1999-16, specimen nos. 151322, 15137, 151339-151340).
Simulium (Trichodagmia)
guianense Wise, 1911 (species complex)
TYPE MATERIAL
GUYANA
PINNED
Lower Rupununi R; 1908, (KS
Wise on label, but actually collected by Melville) - 1
female (man-biting) [LECTOTYPE, head, abdomen, and two legs on slides], 3
females (man-biting) [PARALECTOTYPES, 1 specimen with abdomen and leg on
slide] (BMNH). British Guiana (no locality, but probably the Lower Rupununi
R); 1908, (KS Wise on label, but actually collected by Melville)
- 4 females (man-biting) (PARALECTOTYPES, genitalia of two of the females
on slides) (BMNH, B.M. 1908-207).
SLIDES
Lower Rupununi R; 1908, (KS
Wise on label, but actually collected by Melville) - 1
female (man-biting) (LECTOTYPE, head, abdomen and two legs). Lower Rupununi
River; 1908, (KS Wise on label, but actually collected by Melville)
- 1 female (man-biting) [PARALECTOTYPE, abdomen, wing and hind leg] (BMNH).
British Guiana (no locality, but probably the Lower Rupununi R); 1908, (KS
Wise on label, but actually collected by Melville) - 2
females (man-biting) [PARALECTOTYPES, only genitalia on slide, adults pinned]
(BMNH, B.M. 1908-207).
OTHER MATERIAL
PINNED
Rupununi, Orinduik, R Ireng [site
11, (GUY 17/2, 58/2-5, 59/1-2, 4-5)], 4º44.3'N60º02.3'W, 460 m;
30.xi., 1.xii.1970, (JB Davies) - 3 females, 4males (reared, but not
associated with pupa), 1 female (man-biting) (BMNH, B.M. 1999-16, specimen
nos., 151248, 151249, 151295, 1511300 - 1511303, 151324 - 151327). Rupununi,
Orinduik, R Ireng [site 16, (GUY 16/2, 7, 10)], 4º44.6'N60º02.4'W,
460 m; 21.iii.1970, (JB Davies) - 1 female, 2 males (reared) (BMNH,
B.M. 1999-16, specimen no. 151245 - 47). Rupununi, Orinduik, R Ireng [site
13, (GUY 61/1-3)], 4º44'N60º02.9'W, 450 m; 1.ixi.1970, (JB Davies)
- 1 female (reared) (BMNH, B.M. 1999-16, specimen no. 151277 - 79).
SLIDES
Orinduik, R Tumong, (coll. 74);
2.xii.1970, (JB Davies) - 6 females (man-biting) (BMNH). Rupununi
Orinduik, R Ireng, [site 16, (GUY 16/12)], 4º44.5'N60º02.4'W, 460
m; 21.iii.1970, (JB Davies) - 4 females, 4 males (reared but not associated
with pupae), 2 pupae (BMNH, B.M. 1999-16, specimen no. 151134).
SPIRIT
Orinduik, R Tumong; 2.xii.1970, (JB
Davies) - numerous females (man-biting) (BMNH). Orinduik, R Ireng [site
13, (GUY 61)], 4º44'N60º02.9'W, 450 m; 1.xii.1970, (JB Davies)
- 1 female, 2 males (reared but not associated with pupa), numerous pupae
(BMNH, B.M. 1999-16, sample no. 151152). Rupununi, Orinduik, R Ireng, [site
11, (GUY 58)], 4º44.3'N60º02.3'W, 460 m; 30.xi.1970, (JB Davies)
- numerous pupae (BMNH, B.M. 1999-16, sample no. 151150). Rupununi, Orinduik,
R Ireng, (site 10, (GUY 16/8), 4º44.5'N60º02'W, 460 m; 1.ii.1970,
(JB Davies) - 1 pupa (BMNH, B.M. 1999-16, sample no. 151135). Rupununi,
Orinduik, R Ireng, (site 10, (GUY 16/8), 4º44.5'N60º02'W, 460 m;
21.iii.1970, (JB Davies) - 1 male (reared) (BMNH, B.M. 1999-16, sample
no. 151133). Rupununi, Orinduik, R Ireng, (site 16, (GUY 16/12), 4º44.5'N60º02.4'W,
460 m; 21.iii.1970, (JB Davies) - several females and males (reared
but not associated with pupa), numerous pupae (BMNH, B.M. 1999-16, sample
no. 151134). Rupununi, Orinduik, R Ireng, [site 19, (GUY 19/20)], 4º44.3'N60º02.3'W,
460 m; 22.iii.1970, (JB Davies) - several pupae (BMNH, B.M. 1999-16,
sample no. 151139). Rupununi, Orinduik, R. Ireng, [site 11, (GUY 59/20)],
4º44.3'N60º02.3'W, 460 m; 1.xii.1970, (JB Davies) - numerous
pupae (BMNH, B.M. 1999-16, sample no. 151151).
Simulium (Trichodagmia)
perplexum Shelley, Maia-Herzog, Luna Dias & Couch, 1989
TYPE MATERIAL
GUYANA
PINNED
Kaieteur Falls (Potaro R 5º09'N59º29'W);
1.ix.1937, (OW Richards & Smart) - 1 female [HOLOTYPE, BMNH, B.M.
1937-776]. Same locality and collector as holotype; 1.,4. & 9.ix.1937-
13 females, 11 males [PARATYPES, B.M. 1937-776]. Amatuk Falls (Potaro River);
31.viii.1937, (OW Richards & Smart) - 1 female [PARATYPE, BMNH,
B.M. 1937-776]. Warratuk Falls (Potaro River); 31.viii.1937, (OW Richards & Smart)
- 3 females [PARATYPE, BMNH, B.M. 1937-776].
SLIDES
Kaieteur Falls (Potaro R, 5º09'N59º29'W);
1., 9.ix.1937, (OW Richards & Smart) - 3 females, 3 males (only
abdomen, legs and genitalia) [PARATYPES, BMNH, B.M. 1937-776]. Kaieteur,
savannah; 6.ix.1937, (Richards and Smart) - 1 female (man-biting)
(BMNH, B.M. 1937-776).
ACKNOWLEDGEMENTS
JBD wishes to express
his gratitude for the help and companionship provided by those he encountered
during his main trips to the Rupununi District in 1970 and 1971. In particular,
Mr JA Sankey, British High Commission, and his staff; MH Ali, L Frenker,
and Dr Egbert, Ministry of Health; C McA Ashley, Chief Development Officer,
R Barnwell, K Chung, and S Sagar, District Commissioners; L Lowenfield, FM
Wills, and R Dookah, Field Officers, Malaria Eradication Programme. In Kato,
the hospitality provided by the store owner Sr Brazão was especially
welcome. During a last visit in 1975, assistance was provided by Dr P Munroe,
I Rambajan, as well as S Hingwan of CAREC, Trinidad. We are grateful to Dr
Peter Adler of CUAC, US, Dr Neusa Hamada of Inpa, Brazil, and Dr P Reiter
of IP, France, for the loan of specimens.
REFERENCES
- Coscarón
S 1990. Taxonomía y distribución del subgénero Simulium
(Ectemnaspis) Enderlein (Simuliidae, Diptera, Insecta). Iheringia
sér Zool 70: 109-170.
- Coscarón
S 1991. Fauna de Agua Dulce de la República Argentina. 38. Insecta,
Diptera. 2. Simuliidae. vi+7-304 [+ 67 figures on unnumbered pages],
Fundación para la Educación, la Ciencia y la Cultura, Buenos
Aires.
- Crosskey RW, Howard
TM 1997. A New Taxonomic and Geographical Inventory of World Blackflies (Diptera:
Simuliidae), The Natural History Museum, London, 144 pp.
- Davies JB 1973.
Kabowra flies of the Rupununi district, Guyana. West Indian Med J 22:
199.
- Fairchild GB1940.
Notes on the Simuliidae of Panama (Dipt., Nematocera). Ann Entomolog Soc
Am 33: 701-719.
- Floch H, Abonnenc
E 1946. Simulidés de la Guyane Française. I. S. Guianense Wise
1911, S. Rorotaense n. sp., S. Maro-niense n. sp. Pub
l'Inst Pasteur de la Guyane et du Territoire de l'Inini 136: 1-20.
- Hamada N, Adler
PH 1998. Taxonomy of the Simulium perflavum species-group (Diptera:Simuliidae)
with des-cription of a new species from Brazil. Insecta Mundi 12:
207-226.
- Hamada N, Adler
PH 1999. Cytotaxonomy of four species in the Simulium perflavum species
group (Diptera:Simuliidae) from Brazilian Amazonia. Syst Entomol 24:
273-288.
- Hamada
N, Fouque F 2001. Black flies (Diptera: Simuliidae) of French Guiana: cytotaxonomy
and preliminary list of species. Mem Inst Oswaldo Cruz 96:
955-959.
- Hamada N, Grillet
ME 2001. Black flies (Diptera: Simuliidae) of the Gran Sabana (Venezuela)
and Pacaraima region (Brazil): Distributional and identification keys for
larvae and pupae. Entomotropica 16: 29-49.
- Humphrys E, Tikasingh
ES, Davies JB 1977. The Simulium blackfly problem in
Guyana, South America [p. 41]. Proceedings of the Symposium "Man-made
lakes and human health", Paramaribo, Suriname, October 23-25, 73
pp.
- Knab F 1915. Some
new Neotropical Simuliidae. Bull Entomol Res 6: 279-282.
- Moraes MAP, Shelley
AJ, Luna Dias APA 1985. Mansonella ozzardi no Território Federal
de Roraima, Brasil. Distribuição e achado de um novo vetor
na área do Rio Surumu. Mem Inst Oswaldo Cruz 80: 395-400.
- Nathan MB, Tikasingh
ES, Munroe P 1982. Filariasis in Amerindians of western Guyana with
observations on transmission of Mansonella ozzardi by a Simulium species
of the amazonicum group. Tropenmed Parasit 33: 219-222.
- Py-Daniel V 1982.
Prosimuliini (Diptera:Simuliidae) Neotropical 1- Kempfsimulium V.
Py-Daniel & J. A. Nunes de Mello n.gen; K. simplicicolor (Lutz,
1910) n. comb.; Lutsimulium flavopubescens (Lutz, 1910) n. comb.; Mayacnephia
muzquicensis (Díaz Nájera, 1971) n. comb. Amazoniana 7:
293-333.
- Py-Daniel V 1989.
Novas sinonímias e correções em Simulium com
a revalidação de S. pruinosum Lutz, 1904 (Culicimor-pha,
Simuliidae). Rev Saúde Públ São Paulo 23:
254-257.
- Py-Daniel V, Shelley
AJ 1980. Revisão do Simulium spini-branchium Lutz, 1910 (Diptera:Simuliidae),
com a primeira descrição dos adultos e larva, e redescrição
da pupa. Acta Amazonica 10: 213-223.
- Rambajan I 1979.
A new species of Simulium (Simuliidae: Diptera) from the Lethem area,
Rupununi District, Guyana, South America. J Guyana Museum and Zoo, South
America 45: 63-77.
- Rambajan I 1981a.
Abate for the control of blackfly larvae in the river Burru, Lethem area,
Rupunini District, Guyana. Trop Geogr Med 33: 55-57.
- Rambajan I 1981b.
Some observations on the biting habits of S. incrustatum and S.
amazonicum complex in the Lethem area, Rupunini district, Guyana, South
America. Trop Geogr Med 33: 58-60.
- Ramírez
Pérez J 1983. Los Jejenes de Venezuela, Simposio de oncocercosis
americana. Caicet. Puerto Ayacucho, 15-17 Octubre 1983, 156 pp.
- Ramírez
Pérez J, Vulcano MA 1973. Descripción y re-descripciones de
algunos simúlidos de Venezuela (Diptera: Simuliidae). Arch Ven
Med Trop Parasitol Méd 5: 375-399.
- Sawyer J 1991. A
Comparison of Taxonomic Techniques in the Identification of Sibling Species
of South American Simuliidae, PhD Thesis, University of Salford, UK,
216 pp.
- Scarpassa MV, Hamada
N 2003. Isozyme variation in four species of the Simulium perflavum species
group (Diptera: Simuliidae) from the Brazilian Amazon. Gen Mol Biol 26:
39-46.
- Shelley
AJ, Coscarón S 2001. Simuliid blackflies (Diptera: Simuliidae) and
ceratopogonid midges (Diptera: Cerato-pogonidae) as vectors of Mansonella
ozzardi (Nematoda: Onchocercidae) in Northern Argentina. Mem Inst
Oswaldo Cruz 96: 451-458.
- Shelley AJ, Arzube
M, Couch CA 1989a. The Simuliidae (Diptera) of the Santiago onchocerciasis
focus in Ecuador. Bull Brit Mus Nat Hist (Entomol series) 58:
79-130.
- Shelley AJ, Hernández
LM, Penn M 2002. A biosystematic revision of the blackflies (Diptera: Simuliidae)
of Belize, Central America. Bull Nat Hist Mus London (Entomol series) 71:
135-271.
- Shelley AJ, Lowry
CA, Maia-Herzog M, Luna Dias APA, Moraes MAP 1997. Biosystematic studies
on the Simuli-idae (Diptera) of the Amazonia onchocerciasis focus of Brazil. Bull
Nat Hist Mus London (Entomol series) 66:
1-121.
- Shelley AJ, Luna
Dias APA, Maia-Herzog M 1984. New specific synonymy in Neotropical Simulium
s.l. (Diptera: Simuliidae). Mem Inst Oswaldo Cruz 79: 143-161.
- Shelley AJ, Luna
Dias APA, Moraes MAP, Procunier WS 1987. The status of Simulium oyapockense and S.
limbatum as vectors of human onchocerciasis in Brazilian Amazonia. Med
Vet Parasitol 1: 219-234.
- Shelley AJ, Maia-Herzog
M, Lowry CA, Luna Dias APA, Garritano PR, Shelley A, Camargo M, Carter HG
2000. The Simuliidae (Diptera) of the secondary onchocerciasis focus at Minaçu
in central Brazil. Bull Brit Mus Nat Hist Entomol 69: 171-221.
- Shelley AJ, Maia-Herzog
M, Luna Dias APA, Couch CA 1989b. Description of the adults and pupa of Simulium
(Tri-chodagmia) perplexum, new species (Diptera: Simuliidae). Mem
Inst Oswaldo Cruz 84: 343-349.
- Smart J 1940. Simuliidae
(Dipt.) from British Guiana and the Lesser Antilles. Trans R Entomol Soc
London 90: 1-11.
- Vulcano MA 1981.
Simuliidae. In SH Hurlbert, G Rodriguez, N Dias dos Santos (eds),
Aquatic Biota of Tropical South America. Part 1. Arthropoda, San Diego
State University, California, p. 275-285.
- Wise KS 1911.The
Simuliidae of British Guiana. Timehri 1: 248-254.
Copyright 2004 Instituto Oswaldo Cruz - Fiocruz.
The following images related to this document are available:
Photo images
[oc04101f60-64.jpg]
[oc04101f55-59.jpg]
[oc04101f38-48.jpg]
[oc04101f.jpg]
[oc04101f85-92.jpg]
[oc04101f65-77.jpg]
[oc04101f1-7.jpg]
[oc04101f109-116.jpg]
[oc04101f8-14.jpg]
[oc04101f15-25.jpg]
[oc04101f26-37.jpg]
[oc04101f49-54.jpg]
[oc04101f96-101.jpg]
[oc04101f78-84.jpg]
[oc04101f93-95.jpg]
[oc04101f102-108.jpg]
|
